StaurosporineProtein kinase inhibitor,potent and cell permeable CAS# 62996-74-1 |
- Fasudil
Catalog No.:BCC5262
CAS No.:103745-39-7
- Ro 31-8220
Catalog No.:BCC4295
CAS No.:125314-64-9
- Go 6976
Catalog No.:BCC3703
CAS No.:136194-77-9
- Sotrastaurin (AEB071)
Catalog No.:BCC3857
CAS No.:425637-18-9
- Saikosaponin B2
Catalog No.:BCN5916
CAS No.:58316-41-9
- K-252c
Catalog No.:BCC3706
CAS No.:85753-43-1
Quality Control & MSDS
Number of papers citing our products

Chemical structure
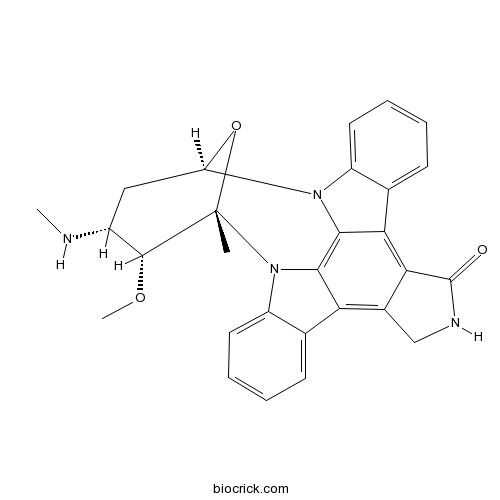
3D structure
| Cas No. | 62996-74-1 | SDF | Download SDF |
| PubChem ID | 44259 | Appearance | Powder |
| Formula | C28H26N4O3 | M.Wt | 466.53 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Synonyms | Antibiotic AM-2282; STS; AM-2282 | ||
| Solubility | DMSO : 125 mg/mL (267.94 mM; Need ultrasonic) H2O : < 0.1 mg/mL (insoluble) | ||
| SMILES | CC12C(C(CC(O1)N3C4=CC=CC=C4C5=C6C(=C7C8=CC=CC=C8N2C7=C53)CNC6=O)NC)OC | ||
| Standard InChIKey | HKSZLNNOFSGOKW-FYTWVXJKSA-N | ||
| Standard InChI | InChI=1S/C28H26N4O3/c1-28-26(34-3)17(29-2)12-20(35-28)31-18-10-6-4-8-14(18)22-23-16(13-30-27(23)33)21-15-9-5-7-11-19(15)32(28)25(21)24(22)31/h4-11,17,20,26,29H,12-13H2,1-3H3,(H,30,33)/t17-,20-,26-,28+/m1/s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Broad spectrum protein kinase inhibitor. Enzymes inhibited include protein kinase C (IC50 = 3 nM), protein kinase A (IC50 = 7 nM), p60v-src tyrosine protein kinase (IC50 = 6 nM) and CaM kinase II (IC50 = 20 nM). |

Staurosporine Dilution Calculator

Staurosporine Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 2.1435 mL | 10.7174 mL | 21.4348 mL | 42.8697 mL | 53.5871 mL |
| 5 mM | 0.4287 mL | 2.1435 mL | 4.287 mL | 8.5739 mL | 10.7174 mL |
| 10 mM | 0.2143 mL | 1.0717 mL | 2.1435 mL | 4.287 mL | 5.3587 mL |
| 50 mM | 0.0429 mL | 0.2143 mL | 0.4287 mL | 0.8574 mL | 1.0717 mL |
| 100 mM | 0.0214 mL | 0.1072 mL | 0.2143 mL | 0.4287 mL | 0.5359 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
Staurosporine, an alkaloid produced in Streptomyces staurospores originally as an antifungal agent, is an inhibitor of a broad spectrum of protein kinases, including protein kinase C (PKC), Camp-dependent protein kinase (PKA), phosphorylase kinase, ribosomal protein S6 kinase, epidermal growth factor receptor (EGF-R) kinase and Ca2+/calmodulin-dependent protein kinase II (Ca/CaM PKII). The inhibition potency is strongest for PKC (IC50 = 2.7 nM) but several-fold lower for other protein kinases. Staurosporine exhibits a strong cytotoxicity to some mammalian tumor cell lines, induces cell apoptosis, and arrests fission yeast cell elongation specifically at a stage immediately after cell division.
Reference
Takashi Toda, Mizuki Shimanuki, and Mitsuhiro Yanagida. Fission yeast genes that confer resistance to staurosporine encode an AP-1-like transcription factor and a protein kinase related to the mammalian ERK1/MAP2 and budding yeast FUS3 and KSS1 kinases. Genes Dev. 1991 5: 60-73
Michelle M. Hill, Mirjana Andelkovic, Derek P. Brazil, Stefano Ferrari, Doriano Fabbro, and Brian A. Hemmings. Insulin-stimulated protein kinase B phosphorylation on Ser-473 is independent of its activity and occurs through a Staurosporine-insensitive kinase. J. Biol. Chem 2001, 276: 25643-25646.
Flavio Meggio, Arianna Donella Deana, Maria Ruzzene, Anna M. Brunati, Luca Cesaro, Barbara Guerra, Thomas Meyer, Helmut Mett, Doriano Fabbro, Pascal Furet, Grazyna Dobrowolska, and Lorenzo A. Pinna. Different susceptibility of protein kinases to staurosporine inhibition kinetic studies and molecular bases for the resistance of protein kinase CK2. Eur. J. Biochem. 234, 317-322 (1995)
- 6-Aminoquinoxaline
Catalog No.:BCC8767
CAS No.:6298-37-9
- XL335
Catalog No.:BCC4501
CAS No.:629664-81-9
- Boc-Tle-OH
Catalog No.:BCC3343
CAS No.:62965-35-9
- Gnetucleistol D
Catalog No.:BCN3400
CAS No.:629643-26-1
- Gomisin G
Catalog No.:BCN2269
CAS No.:62956-48-3
- Gomisin F
Catalog No.:BCN3625
CAS No.:62956-47-2
- (2-Benzothiazolylthio)acetic acid
Catalog No.:BCC8387
CAS No.:6295-57-4
- 6-Methoxy-4-methylcoumarin
Catalog No.:BCN6537
CAS No.:6295-35-8
- Morusinol
Catalog No.:BCN4168
CAS No.:62949-93-3
- Mulberrin
Catalog No.:BCN4167
CAS No.:62949-79-5
- Kuwanon A
Catalog No.:BCN2944
CAS No.:62949-77-3
- H-D-Pro-NH2
Catalog No.:BCC3024
CAS No.:62937-45-5
- Androstenedione
Catalog No.:BCC8296
CAS No.:63-05-8
- Primaquine Diphosphate
Catalog No.:BCC4706
CAS No.:63-45-6
- H-Met-OH
Catalog No.:BCC2993
CAS No.:63-68-3
- Sulfanilamide
Catalog No.:BCC4858
CAS No.:63-74-1
- L-Phenylalanine
Catalog No.:BCN3818
CAS No.:63-91-2
- Phenoxybenzamine HCl
Catalog No.:BCC4334
CAS No.:63-92-3
- Nonacosane
Catalog No.:BCC9102
CAS No.:630-03-5
- Ouabain
Catalog No.:BCC5069
CAS No.:630-60-4
- Phenytoin sodium
Catalog No.:BCC5071
CAS No.:630-93-3
- Corynoxeine
Catalog No.:BCN5002
CAS No.:630-94-4
- Neoprzewaquinone A
Catalog No.:BCN4169
CAS No.:630057-39-5
- MRS 2500 tetraammonium salt
Catalog No.:BCC5881
CAS No.:630103-23-0
Reactive Oxygen Species Evoked by Potassium Deprivation and Staurosporine Inactivate Akt and Induce the Expression of TXNIP in Cerebellar Granule Neurons.[Pubmed:28367274]
Oxid Med Cell Longev. 2017;2017:8930406.
The reactive oxygen species (ROS) play a critical role in neuronal apoptosis; however, the mechanisms are not well understood. It has been shown that thioredoxin-interacting protein (TXNIP) overexpression renders cells more susceptible to oxidative stress and promotes apoptosis and that the activation of PI3K/Akt pathway leads to a downregulation of TXNIP. Here, we evaluated the role of ROS in the regulation of Akt activity and the subsequent regulation of the TXNIP expression in a model of apoptotic death of cerebellar granule neurons (CGN). We observed that two apoptotic conditions that generate ROS at short times led to an increase in the expression of TXNIP in a time-dependent manner; antioxidants significantly reduced this expression. Also, H2O2 caused an increase in TXNIP expression. Moreover, apoptotic conditions induced inactivation of Akt in a time-dependent manner similar to TXNIP expression and H2O2 treatment led to Akt inactivation. Besides, the pharmacological inhibition of Akt increases TXNIP expression and induces CGN cell death. Together, these results suggest that ROS promote neuronal apoptosis through the Akt-TXNIP signaling pathway, supporting the idea that the PI3K/Akt pathway regulates the TXNIP expression. This study highlights the potential importance of this mechanism in neuronal death.
Staurosporine suppresses survival of HepG2 cancer cells through Omi/HtrA2-mediated inhibition of PI3K/Akt signaling pathway.[Pubmed:28349827]
Tumour Biol. 2017 Mar;39(3):1010428317694317.
Staurosporine, which is an inhibitor of a broad spectrum of protein kinases, has shown cytotoxicity on several human cancer cells. However, the underlying mechanism is not well understood. In this study, we examined whether and how this compound has an inhibitory action on phosphatidylinositol 3-kinase (PI3K)/Akt pathway in vitro using HepG2 human hepatocellular carcinoma cell line. Cell viability and apoptosis were determined using 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay and terminal deoxyribonucleotidyl transferase-mediated dUTP-digoxigenin nick end labeling (TUNEL) assay, respectively. Glutathione S-transferase (GST) pull-down assay and co-immunoprecipitation were performed to detect protein-protein interactions. Small interfering RNA (siRNA) was used to silence the expression of targeted protein. We found that Staurosporine significantly decreased cell viability and increased cell apoptosis in a concentration- and time-dependent manner in HepG2 cancer cells, along with the decreased expressions of PDK1 protein and Akt phosphorylation. Staurosporine was also found to enhance Omi/HtrA2 release from mitochondria. Furthermore, Omi/HtrA2 directly bound to PDK1. Pharmacological and genetic inhibition of Omi/HtrA2 restored protein levels of PDK1 and protected HepG2 cancer cells from Staurosporine-induced cell death. In addition, Staurosporine was found to activate autophagy. However, inhibition of autophagy exacerbated cell death under concomitant treatment with Staurosporine. Taken together, our results indicate that Staurosporine induced cytotoxicity response by inhibiting PI3K/Akt signaling pathway through Omi/HtrA2-mediated PDK1 degradation, and the process provides a novel mechanism by which Staurosporine produces its therapeutic effects.
Staurosporine Induces Filamentation in the Human Fungal Pathogen Candida albicans via Signaling through Cyr1 and Protein Kinase A.[Pubmed:28261668]
mSphere. 2017 Mar 1;2(2). pii: mSphere00056-17.
Protein kinases are key regulators of signal transduction pathways that participate in diverse cellular processes. In fungal pathogens, kinases regulate signaling pathways that govern drug resistance, stress adaptation, and pathogenesis. The impact of kinases on the fungal regulatory circuitry has recently garnered considerable attention in the opportunistic fungal pathogen Candida albicans, which is a leading cause of human morbidity and mortality. Complex regulatory circuitry governs the C. albicans morphogenetic transition between yeast and filamentous growth, which is a key virulence trait. Here, we report that Staurosporine, a promiscuous kinase inhibitor that abrogates fungal drug resistance, also influences C. albicans morphogenesis by inducing filamentation in the absence of any other inducing cue. We further establish that Staurosporine exerts its effect via the adenylyl cyclase Cyr1 and the cyclic AMP (cAMP)-dependent protein kinase A (PKA). Strikingly, filamentation induced by Staurosporine does not require the known upstream regulators of Cyr1, Ras1 or Pkc1, or effectors downstream of PKA, including Efg1. We further demonstrate that Cyr1 is capable of activating PKA to enable filamentation in response to Staurosporine through a mechanism that does not require degradation of the transcriptional repressor Nrg1. We establish that Staurosporine-induced filamentation is accompanied by a defect in septin ring formation, implicating cell cycle kinases as potential Staurosporine targets underpinning this cellular response. Thus, we establish Staurosporine as a chemical probe to elucidate the architecture of cellular signaling governing fungal morphogenesis and highlight the existence of novel circuitry through which the Cyr1 and PKA govern a key virulence trait. IMPORTANCE The impact of fungal pathogens on human health is devastating. One of the most pervasive fungal pathogens is Candida albicans, which kills ~40% of people suffering from bloodstream infections. Treatment of these infections is extremely difficult, as fungi are closely related to humans, and there are limited drugs that kill the fungus without host toxicity. The capacity of C. albicans to transition between yeast and filamentous forms is a key virulence trait. Thus, understanding the genetic pathways that regulate morphogenesis could provide novel therapeutic targets to treat C. albicans infections. Here, we establish the small molecule Staurosporine as an inducer of filamentous growth. We unveil distinct regulatory circuitry required for Staurosporine-induced filamentation that appears to be unique to this filament-inducing cue. Thus, this work highlights the fact that small molecules, such as Staurosporine, can improve our understanding of the pathways required for key virulence programs, which may lead to the development of novel therapeutics.
Stathmin decreases cholangiocarcinoma cell line sensitivity to staurosporine-triggered apoptosis via the induction of ERK and Akt signaling.[Pubmed:28178656]
Oncotarget. 2017 Feb 28;8(9):15775-15788.
Cholangiocarcinoma is a rare, but highly fatal malignancy. However, the intrinsic mechanism involved in its tumorigenesis remains obscure. An urgent need remains for a promising target for cholangiocarcinoma biological therapies. Based on comparative proteomical technologies, we found 253 and 231 different spots in gallbladder tumor cell lines and cholangiocarcinoma cell lines, respectively, relative to non-malignant cells. Using Mass Spectrometry (MS) and database searching, we chose seven differentially expressed proteins. High Stathmin expression was found in both cholangiocarcinoma and gallbladder carcinoma cells. Stathmin expression was validated using immunohistochemistry and western blot in cholangiocarcinoma tissue samples and peritumoral tissue. It was further revealed that high Stathmin expression was associated with the repression of Staurosporine-induced apoptosis in the cholangiocarcinoma cell. Moreover, we found that Stathmin promoted cancer cell proliferation and inhibited its apoptosis through protein kinase B (Akt) and extracellular signal-regulated kinase (ERK) signaling. Integrin, beta1 appears to serve as a partner of Stathmin induction of ERK and Akt signaling by inhibiting apoptosis in the cholangiocarcinoma cell. Understanding the regulation of anti-apoptosis effect by Stathmin might provide new insight into how to overcome therapeutic resistance in cholangiocarcinoma.
Staurosporine: an effective inhibitor for Ca2+/calmodulin-dependent protein kinase II.[Pubmed:1846174]
J Neurochem. 1991 Jan;56(1):294-8.
We investigated the effect of Staurosporine on Ca2+/calmodulin-dependent protein kinase II (CaM kinase II) purified from rat brain. (a) Staurosporine (10-100 nM) inhibited the activity of CaM kinase II. The half-maximal and maximal inhibitory concentrations were 20 and 100 nM, respectively. (b) The inhibition with Staurosporine was of the noncompetitive type with respect to ATP, calmodulin, and phosphate acceptor (beta-casein). (c) Staurosporine suppressed the auto-phosphorylation of alpha- and beta-subunits of CaM kinase II at concentrations similar to those at which the enzyme activity was inhibited. (d) Staurosporine also attenuated the Ca2+/calmodulin-independent activity of the autophosphorylated CaM kinase II. These results suggest that Staurosporine inhibits CaM kinase II by interacting with the catalytic domain, distinct from the ATP-binding site or substrate-binding site, of the enzyme and that Staurosporine is an effective inhibitor for CaM kinase II in the cell system.
Staurosporine, a potent inhibitor of phospholipid/Ca++dependent protein kinase.[Pubmed:3457562]
Biochem Biophys Res Commun. 1986 Mar 13;135(2):397-402.
Staurosporine, microbial alkaloid which has been known to have antifungal activity was found to inhibit markedly phospholipid/Ca++dependent protein kinase (protein kinase C) from rat brain, with an IC50 value of 2.7 nM. However, it had little effect on the binding of 3H-phorbol-12, 13-dibutyrate (PDBu) to protein kinase C. The inhibition of protein kinase C was not competitive with phospholipid. This compound also showed the strong cytotoxic effect on the growth of HeLa S3 cells, with an IC50 value of 4 X 10(-12)M under the condition of 72 hr-exposure.


