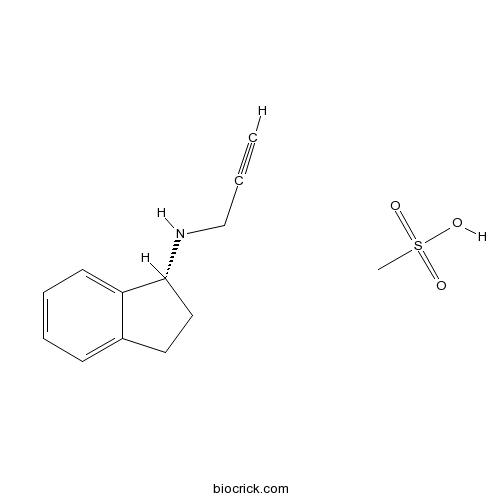
Rasagiline mesylateIrreversible MAO-B inhibitor CAS# 161735-79-1 |
- Safinamide Mesylate
Catalog No.:BCC2320
CAS No.:202825-46-5
- Moclobemide (Ro 111163)
Catalog No.:BCC2322
CAS No.:71320-77-9
Quality Control & MSDS
Number of papers citing our products

Chemical structure

3D structure
| Cas No. | 161735-79-1 | SDF | Download SDF |
| PubChem ID | 3052775 | Appearance | Powder |
| Formula | C13H17NO3S | M.Wt | 267.34 |
| Type of Compound | Miscellaneous | Storage | Desiccate at -20°C |
| Synonyms | AGN1135; TVP1012 | ||
| Solubility | H2O : 25 mg/mL (93.51 mM; Need ultrasonic) | ||
| Chemical Name | methanesulfonic acid;(1R)-N-prop-2-ynyl-2,3-dihydro-1H-inden-1-amine | ||
| SMILES | CS(=O)(=O)O.C#CCNC1CCC2=CC=CC=C12 | ||
| Standard InChIKey | JDBJJCWRXSVHOQ-UTONKHPSSA-N | ||
| Standard InChI | InChI=1S/C12H13N.CH4O3S/c1-2-9-13-12-8-7-10-5-3-4-6-11(10)12;1-5(2,3)4/h1,3-6,12-13H,7-9H2;1H3,(H,2,3,4)/t12-;/m1./s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Rasagiline mesylate is a potent, selective, non-reversible MAO-B inhibitor, with neuroprotective activities, with anti-Parkinson activity. Rasagiline mesylate exhibits neuroprotective and anti-apoptotic activity against several neurotoxins in cell culture. |
| Targets | MAO | ROS |
| In vitro | Effect of the moist-heat sterilization on fabricated nanoscale solid lipid particles containing rasagiline mesylate.[Pubmed: 25838993 ]Int J Pharm Investig. 2015 Apr-Jun;5(2):87-91.Nanoscale solid lipid particles of Rasagiline mesylate (RM) were fabricated by microemulsion technique. The nanoscale particle must be sterile for intravenous administration, and several approaches are available for sterilization. However, the selection of sterilization technique for the fabricated RM loaded nanoscale solid lipid particles mainly depends on the nature of the drug that needs to be encapsulated and release pattern of the polymer.
Controlled release of rasagiline mesylate promotes neuroprotection in a rotenone-induced advanced model of Parkinson's disease.[Pubmed: 22985602]Int J Pharm. 2012 Nov 15;438(1-2):266-78.Microencapsulation of Rasagiline mesylate (RM) into PLGA microspheres was performed by method A (O/W emulsion) and method B (W/O/W double emulsion). The best formulation regarding process yield, encapsulation efficiency and in vitro drug release was that prepared with method A, which exhibited constant drug release for two weeks (K(0)=62.3 μg/day/20mg microspheres). |
| In vivo | Rasagiline mesylate, a new MAO-B inhibitor for the treatment of Parkinson's disease: a double-blind study as adjunctive therapy to levodopa.[Pubmed: 11575866]Clin Neuropharmacol. 2000 Nov-Dec;23(6):324-30.Rasagiline mesylate (TVP-1012) is a potent, selective, non-reversible MAO-B inhibitor, without the tyramine-potentiating effect and with neuroprotective activities. The benefit of rasagiline as monotherapy in patients with early Parkinson's disease (PD) has already been reported. |
| Structure Identification | Eur J Pharm Biopharm. 2013 Nov;85(3 Pt B):1075-83.Evaluation of different substrates for inkjet printing of rasagiline mesylate.[Pubmed: 23563101]The main goal of the present study was to evaluate applicability of the different model substrates, namely orodispersible films (ODFs), porous copy paper sheets, and water impermeable transparency films (TFs) in preparation of the inkjet-printed drug-delivery systems.
Rasagiline mesylate (RM) was used as a low-dose active pharmaceutical ingredient (API). |

Rasagiline mesylate Dilution Calculator

Rasagiline mesylate Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 3.7406 mL | 18.7028 mL | 37.4056 mL | 74.8111 mL | 93.5139 mL |
| 5 mM | 0.7481 mL | 3.7406 mL | 7.4811 mL | 14.9622 mL | 18.7028 mL |
| 10 mM | 0.3741 mL | 1.8703 mL | 3.7406 mL | 7.4811 mL | 9.3514 mL |
| 50 mM | 0.0748 mL | 0.3741 mL | 0.7481 mL | 1.4962 mL | 1.8703 mL |
| 100 mM | 0.0374 mL | 0.187 mL | 0.3741 mL | 0.7481 mL | 0.9351 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
Rasagiline mesylate is a new MAO-B inhibitor for the treatment of idiopathic Parkinson's disease.
- Tebipenempivoxil
Catalog No.:BCC3861
CAS No.:161715-24-8
- Tebipenem
Catalog No.:BCC5550
CAS No.:161715-21-5
- Z-Ile-Leu-aldehyde
Catalog No.:BCC5591
CAS No.:161710-10-7
- Vincamine
Catalog No.:BCN2606
CAS No.:1617-90-9
- Lupenone
Catalog No.:BCN1717
CAS No.:1617-70-5
- Lupeol acetate
Catalog No.:BCN6893
CAS No.:1617-68-1
- Amentoflavone
Catalog No.:BCN6283
CAS No.:1617-53-4
- 2,3,8-Tri-O-methylellagic acid
Catalog No.:BCN1716
CAS No.:1617-49-8
- Caesalpine B
Catalog No.:BCN7377
CAS No.:1616757-60-8
- Caesalpine A
Catalog No.:BCN7376
CAS No.:1616757-59-5
- Dodovislactone B
Catalog No.:BCN7398
CAS No.:1616683-55-6
- Dodovislactone A
Catalog No.:BCN7399
CAS No.:1616683-54-5
- GLP-1 (9-36) amide
Catalog No.:BCC6001
CAS No.:161748-29-4
- 12-Oxocalanolide A
Catalog No.:BCN4699
CAS No.:161753-49-7
- Palmatrubine
Catalog No.:BCN2647
CAS No.:16176-68-4
- Esomeprazole Sodium
Catalog No.:BCC4376
CAS No.:161796-78-7
- Ethyl 2-(4-hydroxyphenyl)-4-methylthiazole-5-carboxylate
Catalog No.:BCC8969
CAS No.:161797-99-5
- Ethyl 2-(3-formyl-4-hydroxyphenyl)-4-methylthiazole-5-carboxylate
Catalog No.:BCC8968
CAS No.:161798-01-2
- Ethyl 2-(3-cyano-4-hydroxyphenyl)-4-methyl-1,3-thiazole-5-carboxylate
Catalog No.:BCC8966
CAS No.:161798-02-3
- Benzamil
Catalog No.:BCC7674
CAS No.:161804-20-2
- Amprenavir (agenerase)
Catalog No.:BCC3619
CAS No.:161814-49-9
- Talampanel(LY300164)
Catalog No.:BCC6378
CAS No.:161832-65-1
- Segetalin A
Catalog No.:BCC9246
CAS No.:161875-97-4
- Prehelminthosporol
Catalog No.:BCN7447
CAS No.:1619-13-2
Controlled release of rasagiline mesylate promotes neuroprotection in a rotenone-induced advanced model of Parkinson's disease.[Pubmed:22985602]
Int J Pharm. 2012 Nov 15;438(1-2):266-78.
Microencapsulation of Rasagiline mesylate (RM) into PLGA microspheres was performed by method A (O/W emulsion) and method B (W/O/W double emulsion). The best formulation regarding process yield, encapsulation efficiency and in vitro drug release was that prepared with method A, which exhibited constant drug release for two weeks (K(0)=62.3 mug/day/20mg microspheres). Exposure of SKN-AS cells to peroxide-induced oxidative stress (1 mM) resulted in cell apoptosis which was significantly reduced by RM (40.7-102.5 muM) as determined by cell viability, ROS production and DNA fragmentation. Daily doses of rotenone (2 mg/kg) given i.p. to rats for 45 days induced neuronal and behavioral changes similar to those occurring in PD. Once an advanced stage of PD was achieved, animals received RM in saline (1 mg/kg/day) or encapsulated within PLGA microspheres (amount of microspheres equivalent to 15 mg/kg RM given on days 15 and 30). After 45 days RM showed a robust effect on all analytical outcomes evaluated with non-statistically significant differences found between its administration in solution or within microparticles however; with this controlled release system administration of RM could be performed every two weeks thereby making this new therapeutic system an interesting approach for the treatment of PD.
Rasagiline mesylate, a new MAO-B inhibitor for the treatment of Parkinson's disease: a double-blind study as adjunctive therapy to levodopa.[Pubmed:11575866]
Clin Neuropharmacol. 2000 Nov-Dec;23(6):324-30.
Rasagiline mesylate (TVP-1012) is a potent, selective, non-reversible MAO-B inhibitor, without the tyramine-potentiating effect and with neuroprotective activities. The benefit of rasagiline as monotherapy in patients with early Parkinson's disease (PD) has already been reported. To evaluate the safety, tolerability, and clinical effect of rasagiline as adjunctive therapy to levodopa, a multicenter, double-blind, randomized, placebo-controlled, parallel-group study (0.5, 1, and 2 mg/d) was conducted for 12 weeks in 70 patients with PD (mean age, 57.4 y; mean disease duration, 5.7 y; 32 patients had motor fluctuations). A beneficial clinical effect was observed in fluctuating patients treated with rasagiline (all doses), expressed as a decrease in total Unified Parkinson's Disease Rating Scale (UPDRS) score (23.0% vs 8.5% in the placebo group). The treatment effect was still evident 6 weeks after drug discontinuation (in all doses). The safety and tolerability of rasagiline were good. Adverse events were no different than those of patients taking placebo. Almost complete platelet MAO-B inhibition was obtained at all rasagiline doses. This study has demonstrated that rasagiline (up to 2 mg/day) has a good safety profile and a beneficial clinical effect in fluctuating patients with PD when given as an add-on to chronic levodopa therapy.
Evaluation of different substrates for inkjet printing of rasagiline mesylate.[Pubmed:23563101]
Eur J Pharm Biopharm. 2013 Nov;85(3 Pt B):1075-83.
The main goal of the present study was to evaluate applicability of the different model substrates, namely orodispersible films (ODFs), porous copy paper sheets, and water impermeable transparency films (TFs) in preparation of the inkjet-printed drug-delivery systems. Rasagiline mesylate (RM) was used as a low-dose active pharmaceutical ingredient (API). Flexible doses of the drug in a single unit were obtained by printing several subsequent layers on top of the already printed ones, using an off-the-shelf consumer thermal inkjet (TIJ) printer. The produced drug-delivery systems were subjected to microscopic and chemical analysis together with solid-state characterization and content uniformity studies. The results revealed that RM recrystallized on the surface of ODFs and TFs, and the printed crystals were arranged in lines. No drug crystals were detected after printing on the surface of the copy paper due to absorption of the ink into the matrix of the substrate. The best linear correlation between the dose of the drug and the number of the printing layers was obtained for the porous copy paper. The other two substrates showed poor linearity and unacceptable standard deviations of the printed drug substance due to limited absorption of the API ink into the carrier. The shear stress between the substrate, the print head, and the paper feeding rollers caused smearing of the drug that had been surface-deposited during the earlier printing cycles. In conclusion, this study indicates that the edible substrates with absorption properties similar to copy paper are favorable for successful preparation of drug-delivery systems by TIJ printers.
Effect of the moist-heat sterilization on fabricated nanoscale solid lipid particles containing rasagiline mesylate.[Pubmed:25838993]
Int J Pharm Investig. 2015 Apr-Jun;5(2):87-91.
BACKGROUND: Nanoscale solid lipid particles of Rasagiline mesylate (RM) were fabricated by microemulsion technique. The nanoscale particle must be sterile for intravenous administration, and several approaches are available for sterilization. However, the selection of sterilization technique for the fabricated RM loaded nanoscale solid lipid particles mainly depends on the nature of the drug that needs to be encapsulated and release pattern of the polymer. MATERIALS AND METHODS: We have preferred moist heat sterilization, as it is the most convenient and the composition of the carrier and incorporated drug should remain unchanged and the incorporated drug should not leak out of the drug carrier. The physical and chemical stability of RM loaded nanoscale solid lipid particles investigated during sterilization and to determine the average mean particle size, polydispersity index, zeta potential (ZP), transmission electron microscopy (TEM), entrapment efficiency (EE), and drug content after autoclaving. RESULT: There were no significant changes in the average mean particle size, polydispersity index, ZP, TEM, EE, and drug content of RM loaded nanoscale solid lipid particles after autoclaving (121 degrees C for 20 min [15 lbs]). CONCLUSION: These observations suggest that the moist heat sterilization by autoclaving is the most suitable method for nanoscale solid lipid formulations.
Pharmacological properties of the anti-Parkinson drug rasagiline; modification of endogenous brain amines, reserpine reversal, serotonergic and dopaminergic behaviours.[Pubmed:12504917]
Neuropharmacology. 2002 Dec;43(7):1110-8.
Rasagiline [N-propargyl-1R(+)-aminoindan; TVP1012] is a potent irreversible monoamine oxidase (MAO) inhibitor with selectivity for type B of the enzyme, which is being developed for treatment of Parkinson's disease. In this study we examined effects of rasagiline on CNS monoamine levels, modification of behavioural response to L-tryptophan, fluoxetine and L-DOPA, and reversal of reserpine syndrome. Reserpine-induced ptosis was reversed by rasagiline at doses above 2 mg x kg(-1) i.p., which inhibit MAO-A as well as MAO-B, but not at MAO-B-selective doses. However, combination of rasagiline (10 mg x kg(-1) i.p.) with L-DOPA or L-tryptophan (50 mg x kg(-1) i.p.), or rasagiline (10 mg x kg(-1) p.o.) with fluoxetine (10 mg x kg(-1) p.o.), did not induce the behavioural hyperactivity syndrome which is seen following inhibition of both MAO-A and MAO-B by tranylcypromine together with the monoamine precursors. Following oral administration, levels of noradrenaline (NA), 5-hydroxytryptamine (5-HT) and dopamine (DA) were unaffected in hippocampus and striatum after single doses of rasagiline up to 2 mg x kg(-1). Following chronic oral administration (21 days, one dose daily), levels of NA, 5-HT and DA in hippocampus and striatum were unaffected by rasagiline at doses up to 1 mg x kg(-1). Rasagiline does not modify CNS monoamine tissue levels or monoamine-induced behavioural syndromes at doses which selectively inhibit MAO-B but not MAO-A.
The anti-Parkinson drug, rasagiline, prevents apoptotic DNA damage induced by peroxynitrite in human dopaminergic neuroblastoma SH-SY5Y cells.[Pubmed:11956966]
J Neural Transm (Vienna). 2002 Apr;109(4):467-81.
Clinical trials for treatment of Parkinson's disease suggest that (-)deprenyl (selegiline), an inhibitor of type B monoamine oxidase, may slow the disease progression. However, the mechanism underlying protection of nigral dopamine neurons by selegiline remains an enigma. Recently, rasagiline, (R)(+)-N-propargyl-1-aminoindan, was reported to be neuroprotective by in vivo and in vitro experiments, which is another selective irreversible inhibitor of type B monoamine oxidase and not metabolized into amphetamine-like derivatives as in the case of selegiline. In this paper, the mechanism of the neuroprotection was examined using human dopaminergic neuroblastoma SH-SY5Y cells against apoptosis induced by peroxynitrite generated from SIN-1. After treatment with SIN-1, the apoptotic DNA damage in the cells was quantified by a single cell gel electrophoresis (comet) assay and by staining with Hoechst 33342. Change in mitochondrial membrane potential, Deltapsim, was measured by use of a fluorescent indicator, JC-1. Rasagiline reduced apoptosis with much more potency than selegiline, and the protection required 20 min pre-incubation before SIN-1 treatment. The protection by rasagiline was proved to be due to stabilization of mitochondrial membrane potential against the collapse induced by SIN-1, whereas rasagiline did not scavenge peroxynitrite directly. The studies on structure-activity relationship showed that a propargylamine group and a hydrophobic group with an adequate intermediate space were required for the protection. These results suggest that rasagiline may protect declining neurons through its anti-apoptotic activity in neurodegenerative diseases.


