PD 180970P210bcr/abl tyrosine kinase inhibitor CAS# 287204-45-9 |
- Imatinib Mesylate (STI571)
Catalog No.:BCC1115
CAS No.:220127-57-1
- Dasatinib (BMS-354825)
Catalog No.:BCC1281
CAS No.:302962-49-8
- Saracatinib (AZD0530)
Catalog No.:BCC1166
CAS No.:379231-04-6
- Bosutinib (SKI-606)
Catalog No.:BCC1167
CAS No.:380843-75-4
- DPH
Catalog No.:BCC1538
CAS No.:484049-04-9
Quality Control & MSDS
Number of papers citing our products

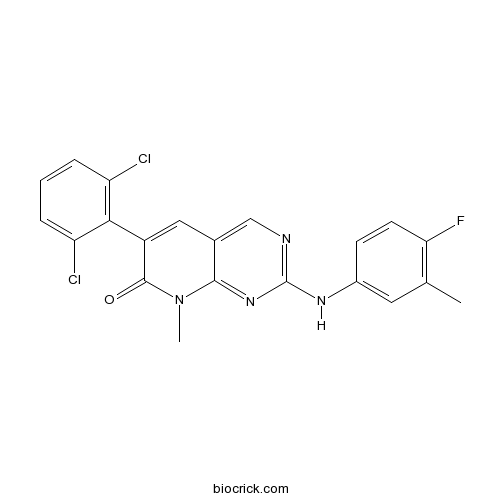
Chemical structure
3D structure
| Cas No. | 287204-45-9 | SDF | Download SDF |
| PubChem ID | 5311104 | Appearance | Powder |
| Formula | C21H15Cl2FN4O | M.Wt | 429.27 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Solubility | Soluble to 50 mM in DMSO | ||
| Chemical Name | 6-(2,6-dichlorophenyl)-2-(4-fluoro-3-methylanilino)-8-methylpyrido[2,3-d]pyrimidin-7-one | ||
| SMILES | CC1=C(C=CC(=C1)NC2=NC=C3C=C(C(=O)N(C3=N2)C)C4=C(C=CC=C4Cl)Cl)F | ||
| Standard InChIKey | SLCFEJAMCRLYRG-UHFFFAOYSA-N | ||
| Standard InChI | InChI=1S/C21H15Cl2FN4O/c1-11-8-13(6-7-17(11)24)26-21-25-10-12-9-14(20(29)28(2)19(12)27-21)18-15(22)4-3-5-16(18)23/h3-10H,1-2H3,(H,25,26,27) | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | ATP-competitive inhibitor of p210bcr/abl tyrosine kinase; selectively induces apoptosis in chronic myeloid leukemia (CML) K562 cells. Inhibits in vivo tyrosine phosphorylation of Gab2, CrkL and p210bcr/abl (IC50 values are 80, 80 and 170 nM respectively). Potently inhibits p210bcr/abl autophosphorylation in vitro (IC50 = 5 nM). Also potently inhibits c-Src and KIT (IC50 values are 0.8 and 50 nM, respectively). |

PD 180970 Dilution Calculator

PD 180970 Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 2.3295 mL | 11.6477 mL | 23.2954 mL | 46.5907 mL | 58.2384 mL |
| 5 mM | 0.4659 mL | 2.3295 mL | 4.6591 mL | 9.3181 mL | 11.6477 mL |
| 10 mM | 0.233 mL | 1.1648 mL | 2.3295 mL | 4.6591 mL | 5.8238 mL |
| 50 mM | 0.0466 mL | 0.233 mL | 0.4659 mL | 0.9318 mL | 1.1648 mL |
| 100 mM | 0.0233 mL | 0.1165 mL | 0.233 mL | 0.4659 mL | 0.5824 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
ATP-competitive inhibitor of p210bcr/abl tyrosine kinase; selectively induces apoptosis in chronic myeloid leukemia (CML) K562 cells. Inhibits in vivo tyrosine phosphorylation of Gab2, CrkL and p210bcr/abl (IC50 values are 80, 80 and 170 nM respectively).
- NCX 4040
Catalog No.:BCC7944
CAS No.:287118-97-2
- Ezatiostat hydrochloride
Catalog No.:BCC4259
CAS No.:286942-97-0
- Fesoterodine Fumarate
Catalog No.:BCC4584
CAS No.:286930-03-8
- 6'-Amino-3',4'-(methylenedioxy)acetophenone
Catalog No.:BCC8760
CAS No.:28657-75-2
- Epoxylathyrol
Catalog No.:BCN5382
CAS No.:28649-60-7
- Euphorbiasteroid
Catalog No.:BCN2781
CAS No.:28649-59-4
- Multicaulisin
Catalog No.:BCN7840
CAS No.:286461-76-5
- L-838,417
Catalog No.:BCC7617
CAS No.:286456-42-6
- Meloscandonine
Catalog No.:BCN5186
CAS No.:28645-27-4
- Nigericin sodium salt
Catalog No.:BCC7915
CAS No.:28643-80-3
- KRN 633
Catalog No.:BCC2544
CAS No.:286370-15-8
- S 14506 hydrochloride
Catalog No.:BCC7174
CAS No.:286369-38-8
- MA 2029
Catalog No.:BCC7983
CAS No.:287206-61-5
- Oxcarbazepine
Catalog No.:BCC5077
CAS No.:28721-07-5
- Scriptaid
Catalog No.:BCC2163
CAS No.:287383-59-9
- 3CAI
Catalog No.:BCC5402
CAS No.:28755-03-5
- Apigenin 5-O-beta-D-glucopyranoside
Catalog No.:BCN5185
CAS No.:28757-27-9
- Rosuvastatin
Catalog No.:BCC4139
CAS No.:287714-41-4
- 4,5,6,7-Tetrahydrothieno [3,2,c]pyridine hydrochloride
Catalog No.:BCC8664
CAS No.:28783-41-7
- Nordihydrocapsaicin
Catalog No.:BCN2387
CAS No.:28789-35-7
- H-Cys(Acm)-OH.HCl
Catalog No.:BCC2903
CAS No.:28798-28-9
- Peptone, bacteriological
Catalog No.:BCC1210
CAS No.:288-88-0
- Tetrazole
Catalog No.:BCC2847
CAS No.:288-94-8
- Heraclenin
Catalog No.:BCN5187
CAS No.:2880-49-1
Pd-Catalyzed Acyl C-O Bond Activation for Selective Ring-Opening of alpha-Methylene-beta-lactones with Amines.[Pubmed:28375015]
Org Lett. 2017 Apr 21;19(8):1966-1969.
A Pd-catalyzed ring-opening of beta-lactones with various types of amines (primary, secondary, and aryl) to provide beta-hydroxy amides with excellent selectivity toward acyl C-O bond cleavage is reported. The utility of this protocol is demonstrated in an asymmetric kinetic resolution providing enantioenriched alpha-methylene-beta-lactones.
CD8 T-cell regulation by T regulatory cells and the programmed cell death protein 1 pathway.[Pubmed:28375543]
Immunology. 2017 Jun;151(2):146-153.
The primary function of the immune system is to protect the host from infectious microorganisms and cancers. However, a major component of the immune response involves the direct elimination of cells in the body and the induction of systemic inflammation, which may result in life-threatening immunopathology. Therefore, the immune system has developed complex mechanisms to regulate itself with a specialized subset of CD4 T lymphocytes (referred to as regulatory T cells) and immune checkpoint pathways, such as the programmed cell death protein 1 pathway. These immune regulatory mechanisms can be exploited by pathogens and tumours to establish persistence in the host, warranting a deeper understanding of how to fine-tune immune responses during these chronic diseases. Here, I discuss various features of immune regulatory pathways and what important aspects must be considered in the next generation of therapies to reverse immune exhaustion, understanding that this process is a natural mechanism to prevent the host from destroying itself.
Pd(II)-Catalyzed Direct ortho-C-H Acylation of Aromatic Ketones by Oxidative Decarboxylation of alpha-Oxocarboxylic Acids.[Pubmed:28374587]
Org Lett. 2017 Apr 21;19(8):2082-2085.
A Pd-catalyzed decarboxylative acylation of aromatic ketones with alpha-oxocarboxylic acids was developed, and 1,2-diacylbenzenes were formed in up to 90% yield with excellent ortho-selectivity. This work demonstrates the first successful attempt to direct C-H acylation of aromatic ketones without the need for prederivatization to imines. The acylation reaction was inhibited by radical scavengers such as TEMPO, and 2,2,6,6-tetramethylpiperidin-1-yl benzoate, the adduct of TEMPO and a benzoyl radical, has been isolated and characterized. This finding is compatible with the intermediacy of acyl radicals. A mechanism involving the reaction of the palladacyclic complexes of aryl ketones with acyl radicals is proposed.
Isocyanide insertion across the Pd-C bond of allenyl and propargyl palladium complexes bearing phosphoquinoline as a spectator ligand. Synthesis of a palladium complex bearing a coordinated cyclobutenyl fragment.[Pubmed:28374876]
Dalton Trans. 2017 Apr 19;46(16):5210-5217.
We have studied the insertion of p-toluenesulfonylmethyl isocyanide (TosMIC) on selected allenyl and propargyl complexes of palladium bearing diphenylphosphine quinoline as a spectator ligand. The fast process gives different products depending on the tautomer involved in the reaction. Thus, the unsubstituted allenyl species yields an insertion complex with the isocyanide coordinated between the metal and the first allenyl carbon. On the other hand, a mixture of phenyl substituted allenyl and propargyl palladium complexes yields a novel species characterized by a cyclo-butenyl fragment directly coordinated to palladium. The solid state structure of such a complex together with an exhaustive kinetic study of the whole process is reported.
Sensitivity of oncogenic KIT mutants to the kinase inhibitors MLN518 and PD180970.[Pubmed:15304388]
Blood. 2004 Dec 1;104(12):3754-7.
Oncogenic mutations of the receptor tyrosine kinase KIT occur in gastrointestinal stromal tumors (GISTs), some cases of acute myelogenous leukemia (AML), and systemic mastocytosis (SM). GISTs commonly contain mutations of the KIT juxtamembrane region while SM and AML harbor active site KIT mutations. Imatinib, which potently inhibits juxtamembrane mutants, is effective for the treatment of GISTs but has no activity against active site mutants. We analyzed the inhibitory potential of 2 small molecule inhibitors, MLN518 and PD180970, against different classes of KIT mutants. Both compounds inhibit the growth of cell lines expressing juxtamembrane mutant KIT. MLN518 additionally targets active site mutant cell lines, inhibiting cell proliferation, KIT, and signal transducer and activator of transcription-3 (Stat3) phosphorylation and inducing apoptosis at concentrations that may be clinically achievable. As phase 1 clinical trials of MLN518 in AML have shown little toxicity, our data suggest MLN518 is a promising candidate for the treatment of SM or AML with KIT mutations.
Activity of the Bcr-Abl kinase inhibitor PD180970 against clinically relevant Bcr-Abl isoforms that cause resistance to imatinib mesylate (Gleevec, STI571).[Pubmed:12499247]
Cancer Res. 2002 Dec 15;62(24):7149-53.
Imatinib mesylate, a selective inhibitor of the Abl tyrosine kinase, is effective as a single-agent therapy for chronic myelogenous leukemia. However, resistance has been reported, particularly in patients with advanced-stage disease. Mutations within the Abl kinase domain are a major cause of resistance, demonstrating that Bcr-Abl remains a critical drug target. Recently, a novel pyrido[2,3-d]pyrimidine derivative, PD180970, has been shown to potently inhibit Bcr-Abl and induce apoptosis in Bcr-Abl-expressing leukemic cells. We analyzed the inhibitory activity of PD180970 against Abl kinase domain mutations and cells expressing clinically relevant mutations. Our data indicate that PD180970 is active against several Bcr-Abl mutations that are resistant to imatinib and support the notion that developing additional Abl kinase inhibitors would be useful as a treatment strategy for chronic myelogenous leukemia.
The pyrido[2,3-d]pyrimidine derivative PD180970 inhibits p210Bcr-Abl tyrosine kinase and induces apoptosis of K562 leukemic cells.[Pubmed:10866298]
Cancer Res. 2000 Jun 15;60(12):3127-31.
PD180970 is a novel pyrido[2,3-d]pyrimidine class of ATP-competitive inhibitor of protein tyrosine kinases. We found that PD180970 inhibited in vivo tyrosine phosphorylation of p210Bcr-Abl (IC50 = 170 nM) and the p210BcrAbl substrates Gab2 and CrkL (IC50 = 80 nM) in human K562 chronic myelogenous leukemic cells. In vitro, PD180970 potently inhibited autophosphorylation of p210Bcr-Abl (IC50 = 5 nM) and the kinase activity of purified recombinant Abl tyrosine kinase (IC50 = 2.2 nM). Incubation of K562 cells with PD180970 resulted in cell death. Results of nuclear staining, apoptotic-specific poly(ADP-ribose) polymerase cleavage, and annexin V binding assays indicated that PD180970 induced apoptosis of K562 cells. In contrast, PD180970 had no apparent effects on the growth and viability of p210Bcr-Abl-negative HL60 human leukemic cells. Thus, PD180970 is among the most potent inhibitors of the p210Bcr-Abl tyrosine kinase, which is present in almost all cases of human chronic myelogenous leukemia. These findings indicate that PD180970 is a promising candidate as a novel therapeutic agent for Bcr-Abl-positive leukemia.


