16 alpha-Hydroxytrametenolic acidCAS# 176390-68-4 |
Quality Control & MSDS
Number of papers citing our products

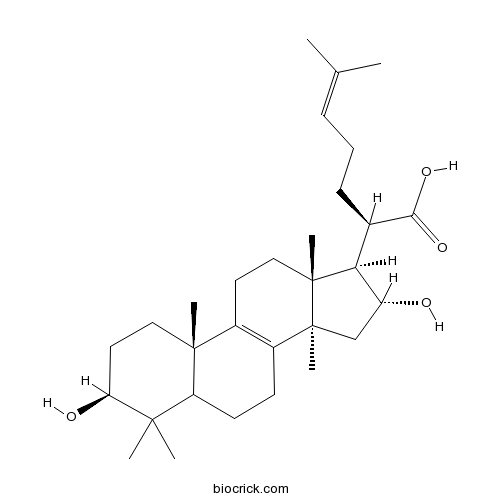
Chemical structure

3D structure
| Cas No. | 176390-68-4 | SDF | Download SDF |
| PubChem ID | 132285301 | Appearance | Powder |
| Formula | C30H48O4 | M.Wt | 472.7 |
| Type of Compound | Triterpenoids | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
| Chemical Name | (2R)-2-[(3S,10S,13R,14R,16R,17R)-3,16-dihydroxy-4,4,10,13,14-pentamethyl-2,3,5,6,7,11,12,15,16,17-decahydro-1H-cyclopenta[a]phenanthren-17-yl]-6-methylhept-5-enoic acid | ||
| SMILES | CC(=CCCC(C1C(CC2(C1(CCC3=C2CCC4C3(CCC(C4(C)C)O)C)C)C)O)C(=O)O)C | ||
| Standard InChIKey | LPHOGVFYEBRXSC-LUXVNGTFSA-N | ||
| Standard InChI | InChI=1S/C30H48O4/c1-18(2)9-8-10-19(26(33)34)25-22(31)17-30(7)21-11-12-23-27(3,4)24(32)14-15-28(23,5)20(21)13-16-29(25,30)6/h9,19,22-25,31-32H,8,10-17H2,1-7H3,(H,33,34)/t19-,22-,23?,24+,25+,28-,29-,30+/m1/s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | 16 alpha-Hydroxytrametenolic acid can inhibit 12-O-tetradecanoylphorbol 13-acetate (TAP)-induced mouse ear edema. |
| Structure Identification | Chem Pharm Bull (Tokyo). 1996 Apr;44(4):847-9.Isolation of inhibitors of TPA-induced mouse ear edema from Hoelen, Poria cocos.[Pubmed: 8681415]
|

16 alpha-Hydroxytrametenolic acid Dilution Calculator

16 alpha-Hydroxytrametenolic acid Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 2.1155 mL | 10.5775 mL | 21.1551 mL | 42.3101 mL | 52.8877 mL |
| 5 mM | 0.4231 mL | 2.1155 mL | 4.231 mL | 8.462 mL | 10.5775 mL |
| 10 mM | 0.2116 mL | 1.0578 mL | 2.1155 mL | 4.231 mL | 5.2888 mL |
| 50 mM | 0.0423 mL | 0.2116 mL | 0.4231 mL | 0.8462 mL | 1.0578 mL |
| 100 mM | 0.0212 mL | 0.1058 mL | 0.2116 mL | 0.4231 mL | 0.5289 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- 16alpha-Hydroxydehydrotrametenolic acid
Catalog No.:BCN1523
CAS No.:176390-66-2
- Fmoc-D-Thr-ol
Catalog No.:BCC2575
CAS No.:176380-53-3
- Dihydropinosylvin methyl ether
Catalog No.:BCN1125
CAS No.:17635-59-5
- D-Raffinose Pentahydrate
Catalog No.:BCN2567
CAS No.:17629-30-0
- Nervogenic acid
Catalog No.:BCN1124
CAS No.:17622-86-5
- LY 354740
Catalog No.:BCC7614
CAS No.:176199-48-7
- Depressine
Catalog No.:BCN7851
CAS No.:176182-06-2
- Maribavir
Catalog No.:BCC5259
CAS No.:176161-24-3
- H-Val-OEt.HCl
Catalog No.:BCC3141
CAS No.:17609-47-1
- Zerumin A
Catalog No.:BCN3684
CAS No.:176050-48-9
- Fucosterol
Catalog No.:BCN6427
CAS No.:17605-67-3
- (±)-Sigmoidin A
Catalog No.:BCN3372
CAS No.:176046-04-1
- Nicotiflorin
Catalog No.:BCN1126
CAS No.:17650-84-9
- 8-Hydroxyodoroside A
Catalog No.:BCN1127
CAS No.:176519-75-8
- Scutebarbatine A
Catalog No.:BCN1128
CAS No.:176520-13-1
- (+)-Taxifolin
Catalog No.:BCN5972
CAS No.:17654-26-1
- Racanisodamine
Catalog No.:BCN8343
CAS No.:17659-49-3
- AAL Toxin TC1
Catalog No.:BCN1736
CAS No.:176590-33-3
- AAL Toxin TC2
Catalog No.:BCN1741
CAS No.:176590-34-4
- AAL Toxin TD1
Catalog No.:BCN1735
CAS No.:176590-35-5
- AAL Toxin TD2
Catalog No.:BCN1740
CAS No.:176590-36-6
- AAL Toxin TE1
Catalog No.:BCN1737
CAS No.:176590-37-7
- AAL Toxin TE2
Catalog No.:BCN1742
CAS No.:176590-38-8
- Urocortin (human)
Catalog No.:BCC5788
CAS No.:176591-49-4
Isolation of inhibitors of TPA-induced mouse ear edema from Hoelen, Poria cocos.[Pubmed:8681415]
Chem Pharm Bull (Tokyo). 1996 Apr;44(4):847-9.
Triterpene carboxylic acids were isolated from the methanol extract of Hoelen, Poria cocos, and found to inhibit 12-O-tetradecanoylphorbol 13-acetate (TAP)-induced mouse ear edema. Their chemical structures were identified as 3 beta,-16 alpha-dihydroxylanosta-7,9(11),24-trien-21-oic acid, 16 alpha-hydroxydehydropachymic acid, 16 alpha-Hydroxytrametenolic acid and dehydrotumulosic acid.


