15,16-Dihydro-15-methoxy-16-oxohardwickiic acidCAS# 115783-35-2 |
Quality Control & MSDS
Number of papers citing our products

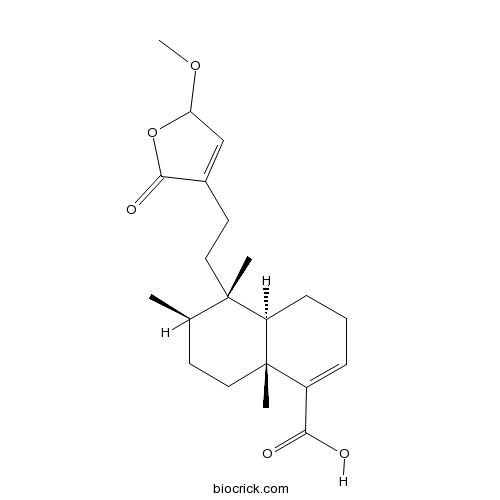
Chemical structure

3D structure
| Cas No. | 115783-35-2 | SDF | Download SDF |
| PubChem ID | 91884914 | Appearance | Powder |
| Formula | C21H30O5 | M.Wt | 362.5 |
| Type of Compound | Diterpenoids | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
| Chemical Name | (4aR,5S,6R,8aR)-5-[2-(2-methoxy-5-oxo-2H-furan-4-yl)ethyl]-5,6,8a-trimethyl-3,4,4a,6,7,8-hexahydronaphthalene-1-carboxylic acid | ||
| SMILES | CC1CCC2(C(C1(C)CCC3=CC(OC3=O)OC)CCC=C2C(=O)O)C | ||
| Standard InChIKey | LKCDRCCSEGFFNK-CULFUZIZSA-N | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||

15,16-Dihydro-15-methoxy-16-oxohardwickiic acid Dilution Calculator

15,16-Dihydro-15-methoxy-16-oxohardwickiic acid Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 2.7586 mL | 13.7931 mL | 27.5862 mL | 55.1724 mL | 68.9655 mL |
| 5 mM | 0.5517 mL | 2.7586 mL | 5.5172 mL | 11.0345 mL | 13.7931 mL |
| 10 mM | 0.2759 mL | 1.3793 mL | 2.7586 mL | 5.5172 mL | 6.8966 mL |
| 50 mM | 0.0552 mL | 0.2759 mL | 0.5517 mL | 1.1034 mL | 1.3793 mL |
| 100 mM | 0.0276 mL | 0.1379 mL | 0.2759 mL | 0.5517 mL | 0.6897 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Galanolactone
Catalog No.:BCN6037
CAS No.:115753-79-2
- 1-O-galloyl-6-O-cinnamoylglucose
Catalog No.:BCN8264
CAS No.:115746-69-5
- 6'-O-beta-D-Glucosylgentiopicroside
Catalog No.:BCN2814
CAS No.:115713-06-9
- Adonifoline
Catalog No.:BCN2056
CAS No.:115712-88-4
- Daidzein dimethyl ether
Catalog No.:BCN6761
CAS No.:1157-39-7
- B2
Catalog No.:BCC7505
CAS No.:115687-05-3
- H-DL-Pro-NH2
Catalog No.:BCC3027
CAS No.:115630-49-4
- Cinnamyl caffeate
Catalog No.:BCN7721
CAS No.:115610-32-7
- Cinnamyl isoferulate
Catalog No.:BCN7718
CAS No.:115610-31-6
- Cinnamyl coumarate
Catalog No.:BCN7739
CAS No.:115610-30-5
- 4-Androstenediol
Catalog No.:BCC8692
CAS No.:1156-92-9
- 2'-Hydroxygenistein
Catalog No.:BCN6036
CAS No.:1156-78-1
- ent-Atisane-3beta,16alpha,17-triol
Catalog No.:BCN6626
CAS No.:115783-44-3
- Tubeimoside II
Catalog No.:BCN2955
CAS No.:115810-12-3
- Tubeimoside III
Catalog No.:BCN2956
CAS No.:115810-13-4
- 2,7-Dideacetoxytaxinine J
Catalog No.:BCN7281
CAS No.:115810-14-5
- Salvianolic acid C
Catalog No.:BCN5376
CAS No.:115841-09-3
- Aurora A Inhibitor I
Catalog No.:BCC2182
CAS No.:1158838-45-9
- N1-Methoxymethyl picrinine
Catalog No.:BCN6038
CAS No.:1158845-78-3
- Tos-Arg-OH
Catalog No.:BCC2873
CAS No.:1159-15-5
- Angoroside C
Catalog No.:BCN4997
CAS No.:115909-22-3
- Tarafenacin D-tartrate
Catalog No.:BCC4148
CAS No.:1159101-48-0
- Salvianolic acid B
Catalog No.:BCN6106
CAS No.:115939-25-8
- Alstonic acid A
Catalog No.:BCN6039
CAS No.:1159579-44-8
Synthesis of apo-13- and apo-15-lycopenoids, cleavage products of lycopene that are retinoic acid antagonists.[Pubmed:28250025]
J Lipid Res. 2017 May;58(5):1021-1029.
Consumption of the tomato carotenoid, lycopene, has been associated with favorable health benefits. Some of lycopene's biological activity may be due to metabolites resulting from cleavage of the lycopene molecule. Because of their structural similarity to the retinoic acid receptor (RAR) antagonist, beta-apo-13-carotenone, the "first half" putative oxidative cleavage products of the symmetrical lycopene have been synthesized. All transformations proceed in moderate to good yield and some with high stereochemical integrity allowing ready access to these otherwise difficult to obtain terpenoids. In particular, the methods described allow ready access to the trans isomers of citral (geranial) and pseudoionone, important flavor and fragrance compounds that are not readily available isomerically pure and are building blocks for many of the longer apolycopenoids. In addition, all of the apo-11, apo-13, and apo-15 lycopenals/lycopenones/lycopenoic acids have been prepared. These compounds have been evaluated for their effect on RAR-induced genes in cultured hepatoma cells and, much like beta-apo-13-carotenone, the comparable apo-13-lycopenone and the apo-15-lycopenal behave as RAR antagonists. Furthermore, molecular modeling studies demonstrate that the apo-13-lycopenone efficiently docked into the ligand binding site of RARalpha. Finally, isothermal titration calorimetry studies reveal that apo-13-lycopenone acts as an antagonist of RAR by inhibiting coactivator recruitment to the receptor.
Coenzyme A thioester formation of 11- and 15-oxo-eicosatetraenoic acid.[Pubmed:28238887]
Prostaglandins Other Lipid Mediat. 2017 May;130:1-7.
Release of arachidonic acid (AA) by cytoplasmic phospholipase A2 (cPLA2), followed by metabolism through cyclooxygenase-2 (COX-2) and 15-hydroxyprostaglandin dehydrogenase (15-PGDH), results in the formation of the eicosanoids 11-oxo- and 15-oxo-eicosatetraenoic acid (oxo-ETE). Both 11-oxo- and 15-oxo-ETE have been identified in human biospecimens but their function and further metabolism is poorly described. The oxo-ETEs contain an alpha,beta-unsaturated ketone and a free carboxyclic acid, and thus may form Michael adducts with a nucleophile or a thioester with the free thiol of Coenzyme A (CoA). To examine the potential for eicosanoid-CoA formation, which has not previously been a metabolic route examined for this class of lipids, we applied a semi-targeted neutral loss scanning approach following arachidonic acid treatment in cell culture and detected inducible long-chain acyl-CoAs including a predominant AA-CoA peak. Interestingly, a series of AA-inducible acyl-CoAs at lower abundance but higher mass, likely corresponding to eicosanoid metabolites, was detected. Using a targeted LC-MS/MS approach we detected the formation of CoA thioesters of both 11-oxo- and 15-oxo-ETE and monitored the kinetics of their formation. Subsequently, we demonstrated that these acyl-CoA species undergo up to four double bond reductions. We confirmed the generation of 15-oxo-ETE-CoA in human platelets via LC-high resolution MS. Acyl-CoA thioesters of eicosanoids may provide a route to generate reducing equivalents, substrates for fatty acid oxidation, and substrates for acyl-transferases through cPLA2-dependent eicosanoid metabolism outside of the signaling contexts traditionally ascribed to eicosanoid metabolites.
Azelaic Acid Topical Formulations: Differentiation of 15% Gel and 15% Foam.[Pubmed:28360967]
J Clin Aesthet Dermatol. 2017 Mar;10(3):37-40. Epub 2017 Mar 1.
In this article, the author reviews topical formulations of azelaic acid used to treat papulopustular rosacea. Emphasis is placed on differences in vehicle technology and potential clinical impact of the possibility for neurosensory cutaneous tolerability reactions.
Clinical impact of galectin-3 in newly diagnosed t (15;17)(q22;q21)/PML-RARa acute promyelocytic leukemia treated with all-trans retinoic acid and arsenic trioxide-based regimens.[Pubmed:28238096]
Ann Hematol. 2017 May;96(5):711-718.
Increased galectin-3 expression has been currently showed to be associated with poor prognosis in some hematological malignancies, such as acute myeloid leukemia, diffuse large B cell lymphoma. However, little is known about the clinical significance of galectin-3 in patients with acute promyelocytic leukemia (APL). We investigated the concentration of serum galectin-3 and characterized the relationship between galectin-3 and outcome in patients with APL. Higher galectin-3 levels were detected in patients with APL compared with the healthy controls (p < 0.001). Higher galectin-3 levels were closely associated with older ages (p < 0.001), the medical history of psoriasis (p = 0.036), coagulopathy (p = 0.042), and CD34 expression (p = 0.004). Compared with patients with lower galectin-3 levels, those with higher galectin-3 levels had significant shorter overall survival (p = 0.028) and relapse-free survival (p = 0.001). Multivariate analysis showed that serum galectin-3 was an independent unfavorable factor for relapse-free survival in patients with APL treated with all-trans retinoic acid and arsenic trioxide-based frontline therapy. Clinical impact of galectin-3 should be further investigated in patients with APL.


