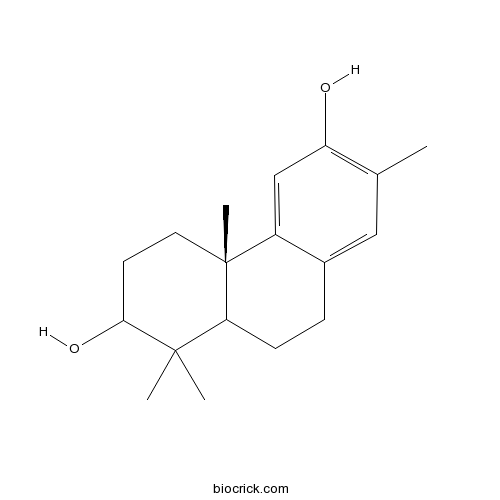
13-Methyl-8,11,13-podocarpatriene-3,12-diolCAS# 769140-74-1 |
Quality Control & MSDS
Number of papers citing our products

Chemical structure

3D structure
| Cas No. | 769140-74-1 | SDF | Download SDF |
| PubChem ID | 71307290 | Appearance | Powder |
| Formula | C18H26O2 | M.Wt | 274.4 |
| Type of Compound | Diterpenoids | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
| Chemical Name | (4aS)-1,1,4a,7-tetramethyl-2,3,4,9,10,10a-hexahydrophenanthrene-2,6-diol | ||
| SMILES | CC1=C(C=C2C(=C1)CCC3C2(CCC(C3(C)C)O)C)O | ||
| Standard InChIKey | ULORBDMEFAYHRJ-LEOMRAHMSA-N | ||
| Standard InChI | InChI=1S/C18H26O2/c1-11-9-12-5-6-15-17(2,3)16(20)7-8-18(15,4)13(12)10-14(11)19/h9-10,15-16,19-20H,5-8H2,1-4H3/t15?,16?,18-/m1/s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||

13-Methyl-8,11,13-podocarpatriene-3,12-diol Dilution Calculator

13-Methyl-8,11,13-podocarpatriene-3,12-diol Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 3.6443 mL | 18.2216 mL | 36.4431 mL | 72.8863 mL | 91.1079 mL |
| 5 mM | 0.7289 mL | 3.6443 mL | 7.2886 mL | 14.5773 mL | 18.2216 mL |
| 10 mM | 0.3644 mL | 1.8222 mL | 3.6443 mL | 7.2886 mL | 9.1108 mL |
| 50 mM | 0.0729 mL | 0.3644 mL | 0.7289 mL | 1.4577 mL | 1.8222 mL |
| 100 mM | 0.0364 mL | 0.1822 mL | 0.3644 mL | 0.7289 mL | 0.9111 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Triacsin C
Catalog No.:BCC7377
CAS No.:76896-80-5
- Bz-Gly-OH.HCl
Catalog No.:BCC2945
CAS No.:7689-50-1
- Camptothecin
Catalog No.:BCN4318
CAS No.:7689-03-4
- Olivil 4'-O-glucoside
Catalog No.:BCN7557
CAS No.:76880-93-8
- 8-Methyl-8-azabicyclo[3.2.1]octane-3,6-diol, 9CI; (3RS,6RS)-form, 3-O-Ac
Catalog No.:BCN1361
CAS No.:7688-76-8
- 6-Hydroxywogonin
Catalog No.:BCN6556
CAS No.:76844-70-7
- Przewaquinone A
Catalog No.:BCN3004
CAS No.:76843-23-7
- 5-BDBD
Catalog No.:BCC7717
CAS No.:768404-03-1
- Famotidine
Catalog No.:BCC4529
CAS No.:76824-35-6
- Danshensu
Catalog No.:BCN8513
CAS No.:76822-21-4
- Pimaricin
Catalog No.:BCN2216
CAS No.:7681-93-8
- Ronidazole
Catalog No.:BCC4840
CAS No.:7681-76-7
- Begacestat
Catalog No.:BCC2346
CAS No.:769169-27-9
- Viscosalactone B
Catalog No.:BCN7945
CAS No.:76938-46-0
- 8-Bromo-cAMP, sodium salt
Catalog No.:BCC8078
CAS No.:76939-46-3
- Onitin 2'-O-glucoside
Catalog No.:BCN4319
CAS No.:76947-60-9
- Cleomiscosin A
Catalog No.:BCN4320
CAS No.:76948-72-6
- DL-alpha-Tocopherylacetate
Catalog No.:BCN2904
CAS No.:7695-91-2
- Kalii Dehydrographolidi Succinas
Catalog No.:BCN8523
CAS No.:76958-99-1
- Nizatidine
Catalog No.:BCC4522
CAS No.:76963-41-2
- H-D-2-Nal-OH.HCl
Catalog No.:BCC3286
CAS No.:76985-09-6
- Boc-D-2-Nal-OH
Catalog No.:BCC3288
CAS No.:76985-10-9
- Cleomiscosin B
Catalog No.:BCN3898
CAS No.:76985-93-8
- TPT-260
Catalog No.:BCC5171
CAS No.:769856-81-7
Characterization of dibenzo[a,l]pyrene-trans-11,12-diol (dibenzo[def,p]chrysene) glucuronidation by UDP-glucuronosyltransferases.[Pubmed:21780761]
Chem Res Toxicol. 2011 Sep 19;24(9):1549-59.
Dibenzo[a,l]pyrene (DB[a,l]P) (dibenzo[def,p]chrysene) is a highly carcinogenic polycyclic aromatic hydrocarbon (PAH) that has been identified in tobacco smoke and is found in our environment due to incomplete combustion of organic matter. Its metabolites are known to form stable DNA adducts in bacteria and mammalian cells, and can lead to tumors in animal models. Glucuronidation of major metabolites of DB[a,l]P by the uridine-5'-diphosphate glucuronosyltransferase (UGT) family of enzymes is an important route of detoxification of this pro-carcinogen. The focus of the current study was to characterize the glucuronidation of the pro-carcinogenic enantiomers DB[a,l]P-(+)-trans-11S,12S-diol and DB[a,l]P-(-)-trans-11R,12R-diol. Glucuronidation assays with HEK293 cell lines overexpressing individual human UGT enzymes demonstrated that UGTs 1A1, 1A4, 1A7, 1A8, 1A9, 1A10, and 2B7 glucuronidated one or both DB[a,l]P-trans-11,12-diol enantiomers. Three glucuronide conjugates were observed in activity assays with UGTs 1A1 and 1A10, while two glucuronides were formed by UGTs 1A7, 1A8, and 1A9, and one glucuronide was made by UGT1A4 and UGT2B7. Enzyme kinetic analysis indicated that UGT1A9 was the most efficient UGT at forming both the (+)-DB[a,l]P-11-Gluc and (-)-DB[a,l]P-11-Gluc products, while UGTs 1A1 and 1A10 were the most efficient at forming the (+)-DB[a,l]P-12-Gluc product (as determined by k(cat)/K(M)). Incubations with human liver microsomes showed the formation of three diastereomeric glucuronide products: (+)-DB[a,l]P-11-Gluc, (+)-DB[a,l]P-12-Gluc, and (-)-DB[a,l]P-11-Gluc, with an average overall ratio of 31:32:37 in four liver specimens. Human bronchus and trachea tissue homogenates demonstrated glucuronidation activity against both DB[a,l]P-trans-11,12-diol enantiomers, with both tissues producing the (+)-DB[a,l]P-11-Gluc and (+)-DB[a,l]P-12-Gluc with little or no formation of (-)-DB[a,l]P-11-Gluc. These results indicate that multiple UGTs are involved in the stereospecific glucuronidation of DB[a,l]P-trans-11,12-diol in a pattern consistent with their expression in respiratory tract tissues and that glucuronidation may be an important first-line detoxification mechanism of DB[a,l]P metabolites.
Facile interstrand migration of the hydrocarbon moiety of a dibenzo[a,l]pyrene 11,12-diol 13,14-epoxide adduct at N2 of deoxyguanosine in a duplex oligonucleotide.[Pubmed:16881636]
J Am Chem Soc. 2006 Aug 9;128(31):10079-84.
When a synthesized deoxyribonucleotide duplex, 5'-CCATCGCTACC-3'.5'-GGTAGCGATGG-3', containing a trans 14R dibenzo[a,l]pyrene (DB[a,l]P) adduct, corresponding to trans opening of the (+)-(11S,12R)-diol (13R,14S)-epoxide by N (2) of the central G residue, was allowed to stand for 2-6 days at ambient temperature in neutral aqueous solution, three new products were observed on denaturing HPLC. One of these corresponded to loss of the DB[a,l]P moiety from the original adducted strand to give an 11-mer with an unmodified central dG. The other two products resulted from a highly unexpected migration of the hydrocarbon moiety to either dG5 or dG7 of the complementary strand, 5'-GGTAG5CG7ATGG-3'. Enzymatic hydrolysis of the two 11-mer migration products followed by CD spectroscopy of the isolated adducted nucleosides indicated that, in both cases, the hydrocarbon moiety had undergone configurational inversion at C14 to give the cis 14S DB[a,l]P dG adduct. MS/MS and partial enzymatic hydrolysis showed that the major 11-mer had the hydrocarbon at dG7. Two 11-mer oligonucleotides were synthesized with a single cis 14S DB[a,l]P dG adduct either at G7 or at G5 and were found to be chromatographically identical to the major and minor migration products, respectively. Although HPLC evidence suggested that a small extent of hydrocarbon migration from the trans 14S DB[a,l]P dG diastereomer also occurred, the very small amount of presumed migration products from this isomer precluded their detailed characterization. This interstrand migration appears unique to DB[a,l]P adducts and has not been observed for their fjord-region benzo[c]phenanthrene or bay-region benzo[a]pyrene analogues.
In vitro metabolism of benzo[a]pyrene-7,8-dihydrodiol and dibenzo[def,p]chrysene-11,12 diol in rodent and human hepatic microsomes.[Pubmed:28119020]
Toxicol Lett. 2017 Mar 5;269:23-32.
Polycyclic aromatic hydrocarbons (PAHs) are contaminants that are ubiquitously found in the environment, produced through combustion of organic matter or petrochemicals, and many of which are procarcinogens. The prototypic PAH, benzo[a]pyrene (B[a]P) and the highly carcinogenic dibenzo[def,p]chrysene (DBC) are metabolically activated by isoforms of the P450 enzyme superfamily producing benzo[a]pyrene-7,8-dihydrodiol (B[a]P diol), dibenzo[def,p]chrysene-11,12 diol (DBC diol). Each of these diols can be further metabolized by cytochrome P450 enzymes to highly reactive diol-epoxide metabolites that readily react with DNA or by phase II conjugation facilitating excretion. To complement prior in vitro metabolism studies with parent B[a]P and DBC, both phase I metabolism and phase II glucuronidation of B[a]P diol and DBC diol were measured in hepatic microsomes from female B6129SF1/J mice, male Sprague-Dawley rats, and female humans. Metabolic parameters, including intrinsic clearance and Michaelis-Menten kinetics were calculated from substrate depletion data. Mice and rats demonstrated similar B[a]P diol phase I metabolic rates. Compared to rodents, human phase I metabolism of B[a]P diol demonstrated lower overall metabolic capacity, lower intrinsic clearance at higher substrate concentrations (>0.14muM), and higher intrinsic clearance at lower substrate concentrations (<0.07muM). Rates of DBC diol metabolism did not saturate in mice or humans and were highest overall in mice. Higher affinity constants and lower capacities were observed for DBC diol glucuronidation compared to B[a]P diol glucuronidation; however, intrinsic clearance values for these compounds were consistent within each species. Kinetic parameters reported here will be used to extend physiologically based pharmacokinetic (PBPK) models to include the disposition of B[a]P and DBC metabolites in animal models and humans to support future human health risk assessments.
Formation and differential repair of covalent DNA adducts generated by treatment of human cells with (+/-)-anti-dibenzo[a,l]pyrene-11,12-diol-13,14-epoxide.[Pubmed:19053321]
Chem Res Toxicol. 2009 Jan;22(1):81-9.
Dibenzo[a,l]pyrene (DBP) is the most potent tumor initiating polycyclic aromatic hydrocarbon tested to date in rodent tumor models. To investigate how DBP adduct formation and removal might influence carcinogenesis, we have examined the effects of treatment of several nucleotide excision repair (NER)-proficient (NER(+)) and -deficient (NER(-)) cell lines with the carcinogenic metabolite (+/-)-anti-DBP-11,12-diol-13,14-epoxide (DBPDE). The treatment of NER(-) cells with (+/-)-anti-DBPDE for 0.5, 1, or 2 h yielded similar total adduct levels, indicating that adduct formation was essentially complete during a 2 h treatment period with no additional adducts produced after replacement of media. In all cell lines, treatment with (+/-)-anti-DBPDE generated five major and at least two minor adducts that were chromatographically identical to those formed by direct treatment of 3'-GMP and 3'-AMP with (+/-)-anti-DBPDE. When adduct levels were assessed in NER(-) cells, the number of adducts/10(9) nucleotides decreased over time, suggesting that DNA replication was ongoing, so we incorporated a normalization strategy based on DNA synthesis. This strategy indicated that DBPDE-DNA adduct levels in NER(-) cells are stable over time. After normalization for DNA synthesis in the NER(+) cells, our data indicated that three adducts showed biphasic repair kinetics. A faster rate of removal was observed during the first 6 h following DBPDE removal followed by a slower rate for up to 34 h. Importantly, two of the major guanine adducts were particularly refractory to removal in the NER(+) cells. Our results suggest that the extreme carcinogenicity of DBPDE may result from the ability of a substantial percentage of two structurally distinct DBPDE-DNA adducts to escape repair.


