12-Oleanene-3,6-diolCAS# 41498-79-7 |
Quality Control & MSDS
Number of papers citing our products

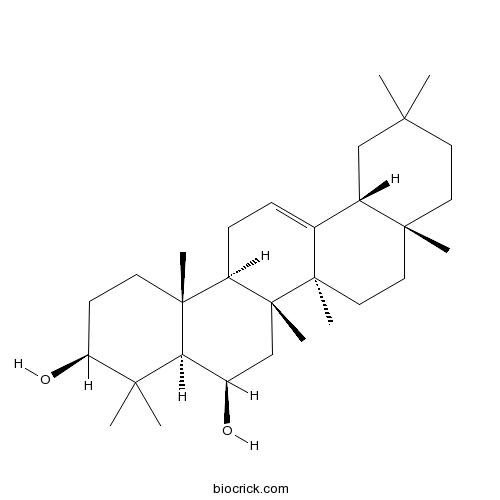
Chemical structure

3D structure
| Cas No. | 41498-79-7 | SDF | Download SDF |
| PubChem ID | 3084830 | Appearance | Cryst. |
| Formula | C30H50O2 | M.Wt | 442.72 |
| Type of Compound | Triterpenoids | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
| Chemical Name | (3S,4aR,5R,6aR,6bS,8aR,12aR,14aR,14bR)-4,4,6a,6b,8a,11,11,14b-octamethyl-1,2,3,4a,5,6,7,8,9,10,12,12a,14,14a-tetradecahydropicene-3,5-diol | ||
| SMILES | CC1(CCC2(CCC3(C(=CCC4C3(CC(C5C4(CCC(C5(C)C)O)C)O)C)C2C1)C)C)C | ||
| Standard InChIKey | JYNBNJRQZZSLPN-NYVWVNPOSA-N | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | 12-Oleanene-3,6-diol has antitumor activity, it can induce apoptosis and has inhibition effect on the proliferation in RKO cell line. |
| In vitro | Antitumor effects of novel triterpene from Celastrus hypoleucus on human colorectal cancer cell line RKO in vitro.[Reference: WebLink]China journal of Chinese materia medica, 2006, 31(17):1450-1453.To investigate the effects of novel triterpene (12-Oleanene-3,6-diol
) from Celastrus hypoleucus on the proliferation and apoptosis of human colorectal cancer cell line RKO. Oleanen induces apoptosis of cervical cancer cells by up-regulation of Bim.[Reference: WebLink]International Journal of Gynecological Cancer, 2012, 22(1):38-42.Plants belonging to the genus Celastrus exhibit antitumor activity and the ability to reverse multidrug resistance in tumor cells; however, it remains unclear whether the compound oleanen from Celastrus hypoleucus also exhibits antitumor activity. The objective of this study was to explore the inhibitory effect of 12-oleanene-3β, 6α-diol (12-Oleanene-3,6-diol, oleanen) on the proliferation of cervical cancer HeLa cells in vitro, as well as its relative mechanism. |

12-Oleanene-3,6-diol Dilution Calculator

12-Oleanene-3,6-diol Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 2.2588 mL | 11.2938 mL | 22.5876 mL | 45.1753 mL | 56.4691 mL |
| 5 mM | 0.4518 mL | 2.2588 mL | 4.5175 mL | 9.0351 mL | 11.2938 mL |
| 10 mM | 0.2259 mL | 1.1294 mL | 2.2588 mL | 4.5175 mL | 5.6469 mL |
| 50 mM | 0.0452 mL | 0.2259 mL | 0.4518 mL | 0.9035 mL | 1.1294 mL |
| 100 mM | 0.0226 mL | 0.1129 mL | 0.2259 mL | 0.4518 mL | 0.5647 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- 3-Amino-1-phenyl-2-pyrazolin-5-one
Catalog No.:BCC8605
CAS No.:4149-06-8
- Belinostat (PXD101)
Catalog No.:BCC2153
CAS No.:414864-00-9
- Turkesterone
Catalog No.:BCN2363
CAS No.:41451-87-0
- Bruceantin
Catalog No.:BCN7618
CAS No.:41451-75-6
- Onetine
Catalog No.:BCN2102
CAS No.:41451-67-6
- Phalaenopsine Is
Catalog No.:BCN2016
CAS No.:41451-64-3
- (+)-N-Methylallosedridine
Catalog No.:BCN5470
CAS No.:41447-16-9
- (-)-N-Methylsedridine
Catalog No.:BCN5469
CAS No.:41447-15-8
- ODQ
Catalog No.:BCC6829
CAS No.:41443-28-1
- 5-Heptadecylresorcinol
Catalog No.:BCN4750
CAS No.:41442-57-3
- 8-Hydroxyapigenin
Catalog No.:BCN8404
CAS No.:41440-05-5
- 5-Benzoylpentanoic acid
Catalog No.:BCC8740
CAS No.:4144-62-1
- Daturaolone
Catalog No.:BCN3904
CAS No.:41498-80-0
- Koaburaside monomethyl ether
Catalog No.:BCN5471
CAS No.:41514-64-1
- 2,6,16-Kauranetriol
Catalog No.:BCN5472
CAS No.:41530-90-9
- 1-(3,4-dimethoxyphenyl)-2-(4-allly-2,6-dimethoxyphenoxy)propan-1-ol
Catalog No.:BCN1445
CAS No.:41535-95-9
- N6-Cyclopentyladenosine
Catalog No.:BCC7160
CAS No.:41552-82-3
- SN-6
Catalog No.:BCC7273
CAS No.:415697-08-4
- RI-1
Catalog No.:BCC1896
CAS No.:415713-60-9
- Poricoic acid G
Catalog No.:BCN8267
CAS No.:415724-84-4
- Carboplatin
Catalog No.:BCC1170
CAS No.:41575-94-4
- 4,R-ajmalicine N-oxide
Catalog No.:BCN5473
CAS No.:41590-29-8
- (-)-Pinoresinol 4-O-glucoside
Catalog No.:BCN7251
CAS No.:41607-20-9
- Ylangenol
Catalog No.:BCN6705
CAS No.:41610-69-9
(1S,2E,4S,7E,11E)-2,7,11-Cembratriene-4,6-diol semisynthetic analogs as novel c-Met inhibitors for the control of c-Met-dependent breast malignancies.[Pubmed:27681240]
Bioorg Med Chem. 2016 Nov 15;24(22):5748-5761.
(1S,2E,4S,6R,7E,11E)-2,7,11-Cembratriene-4,6-diol (1) and its 4-epi-analog (2) are the cembranoid precursors to several key flavor ingredients in most Nicotiana (tobacco) species. Nearly 40-60% of 1 and 2 are purposely degraded during the commercial tobacco fermentation. However, 1 and 2 display promising bioactivities, including anticancer. Breast cancer is the most diagnosed cancer in women and ranked second female disease killer. The receptor tyrosine kinase c-Met correlates with aggressiveness of certain breast cancer phenotypes and thus considered a valid therapeutic target. This study reports the discovery and optimization of the tobacco-based cembranoid 1 as a novel c-Met inhibitory scaffold using combined structure- and ligand-based approaches. 1 displayed antiproliferative, anti-migratory and anti-invasive effects against the c-Met overexpressing MDA-MB-231 breast cancer cells at moderate muM concentrations. The Z'-LYTE kinase platform and Western blot analysis identified c-Met as a potential macromolecular target. Rationally designed carbamate analogs were proposed to probe additional targeted c-Met interactions and improve the cellular potency. The 6-phenyl carbamate 3 showed enhanced c-Met inhibitory activity. Structure-activity relationships of different substituents on the 3's phenyl moiety were studied. The most active analog 20 showed potent in vitro anticancer activity against the MDA-MB-231 breast cancer cells at low muM concentrations, with minimal toxicity on the non-tumorigenic MCF-10A mammary epithelial cells. Cembranoid 20 potently inhibited the c-Met catalytic activity in Z'-LYTE kinase assay and various cellular c-Met-driven signaling pathways. Furthermore, 20 displayed a robust antitumor activity in a breast cancer xenograft athymic mouse model and thus promoted to the lead rank. Cembranoids are novel c-Met inhibitors appropriate for future use to control c-Met dependent malignancies.
Neuroprotective activity of (1S,2E,4R,6R,-7E,11E)-2,7,11-cembratriene-4,6-diol (4R) in vitro and in vivo in rodent models of brain ischemia.[Pubmed:25677097]
Neuroscience. 2015 Apr 16;291:250-259.
(1S,2E,4R,6R,-7E,11E)-2,7,11-cembratriene-4,6-diol (4R) is a precursor to key flavor ingredients in leaves of Nicotiana species. The present study shows 4R decreased brain damage in rodent ischemic stroke models. The 4R-pretreated mice had lower infarct volumes (26.2+/-9.7 mm3) than those in control groups (untreated: 63.4+/-4.2 mm3, DMSO: 60.2+/-14.2 mm3). The 4R-posttreated rats also had less infarct volumes (120+/-65 mm3) than those in the rats of the DMSO group (291+/-95 mm3). The results from in vitro experiments indicate that 4R decreased neuro2a cell (neuroblastoma cells) apoptosis induced by oxygen-glucose deprivation (OGD), and improved the population spikes' (PSs) recovery in rat acute hippocampal slices under OGD; a phosphatidylinositol 3-kinase (PI3K) inhibitor, wortmannin, abolished the effect of 4R on PSs recovery. Furthermore, 4R also inhibited monocyte adhesion to murine brain-derived endothelial (bEND5) cells and upregulation of intercellular adhesion molecule-1(ICAM-1) induced by OGD/reoxygenation (OGD/R), and restored the p-Akt level to pre-OGD/R values in bEND5 cells. In conclusion, the present study indicates that 4R has a protective effect in rodent ischemic stroke models. Inhibition of ICAM-1 expression and restoration of Akt phosphorylation are the possible mechanisms involved in cellular protection by 4R.
The tobacco cembranoid (1S,2E,4S,7E,11E)-2,7,11-cembratriene-4,6-diol as a novel angiogenesis inhibitory lead for the control of breast malignancies.[Pubmed:28583806]
Bioorg Med Chem. 2017 Aug 1;25(15):3911-3921.
(1S,2E,4S,6R,7E,11E)-2,7,11-cembratriene-4,6-diol (1) and its 4-epi-analog (2) are diterpene precursors of the key flavor components in most Nicotiana (tobacco) species that purposely degraded during commercial tobacco fermentation. Angiogenesis, recruitment of new blood vessels, is important for tumor growth, survival and metastasis that can be targeted to control cancer. This study shows evidences and potential of the cembranoid 1 as a potent angiogenesis modulator through targeting VEGFR2. In silico study suggested favorable docking scores and binding affinity of 1 at the ATP binding pocket of VEGFR2. The binding mode of 1 was parallel to the standard FDA-approved antiangiogenic drug sunitinib (4). In vitro, cembranoid 1 significantly reduced the activated VEGFR2 levels in multiple breast cancer cell lines. Intraperitoneal 40mg/kg, 3X/week treatment of 1 significantly reduced the MDA-MB-231 cells breast tumor size in mice. Immunohistochemistry and Western blotting analysis of the treated mice tumors showed significant downregulation of the vasculogenesis marker CD31 and suppressed activated VEGFR2-paxillin-FAK pathway. Matrigel study in Swiss albino mice showed similar trend. The tobacco cembranoid 1 is a potential antiangiogenic lead useful for future use to control breast malignancies.
Design and Synthesis of 2-Alkylpyrimidine-4,6-diol and 6-Alkylpyridine-2,4-diol as Potent GPR84 Agonists.[Pubmed:27326330]
ACS Med Chem Lett. 2016 Mar 30;7(6):579-83.
A series of alkylpyrimidine-4,6-diol derivatives were designed and synthesized as novel GRP84 agonists based on a high-throughput screening (HTS) hit 1. 6-Nonylpyridine-2,4-diol was identified as the most potent agonist of GPR84 reported so far, with an EC50 of 0.189 nM. These novel GPR84 agonists will provide valuable tools for the study of the physiological functions of GPR84.


