11-Deoxyalisol BCAS# 155073-73-7 |
Quality Control & MSDS
Number of papers citing our products

Chemical structure

3D structure
| Cas No. | 155073-73-7 | SDF | Download SDF |
| PubChem ID | 101661274 | Appearance | Powder |
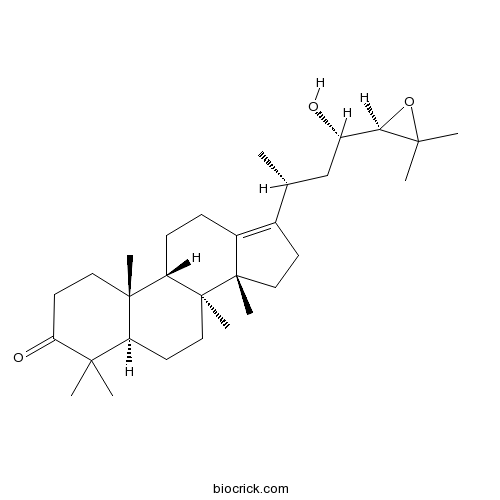
| Formula | C30H48O3 | M.Wt | 456.7 |
| Type of Compound | Triterpenoids | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
| Chemical Name | (5R,8S,9S,10R,14R)-17-[(2R,4S)-4-[(2R)-3,3-dimethyloxiran-2-yl]-4-hydroxybutan-2-yl]-4,4,8,10,14-pentamethyl-1,2,5,6,7,9,11,12,15,16-decahydrocyclopenta[a]phenanthren-3-one | ||
| SMILES | CC(CC(C1C(O1)(C)C)O)C2=C3CCC4C5(CCC(=O)C(C5CCC4(C3(CC2)C)C)(C)C)C | ||
| Standard InChIKey | FRARORPBMYOKKW-DNRQRXMOSA-N | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Structure Identification | Chinese Journal of Pharmaceutical Analysis, 2015,35(2):210-217.Determination of triterpenoids in Rhizoma Alismatis extracts.[Reference: WebLink]To establish ultraviolet-visible(UV-Vis)spectrophotometry and HPLC method for determination of triterpenoids in Rhizoma Alismatis extracts. |

11-Deoxyalisol B Dilution Calculator

11-Deoxyalisol B Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 2.1896 mL | 10.9481 mL | 21.8962 mL | 43.7924 mL | 54.7405 mL |
| 5 mM | 0.4379 mL | 2.1896 mL | 4.3792 mL | 8.7585 mL | 10.9481 mL |
| 10 mM | 0.219 mL | 1.0948 mL | 2.1896 mL | 4.3792 mL | 5.4741 mL |
| 50 mM | 0.0438 mL | 0.219 mL | 0.4379 mL | 0.8758 mL | 1.0948 mL |
| 100 mM | 0.0219 mL | 0.1095 mL | 0.219 mL | 0.4379 mL | 0.5474 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- 24,25-Dihydroxycycloartan-3-one
Catalog No.:BCN1692
CAS No.:155060-48-3
- SM-21 maleate
Catalog No.:BCC6780
CAS No.:155059-42-0
- N-[2-(Piperidinylamino)ethyl]-4-iodobenzamide
Catalog No.:BCC6784
CAS No.:155054-42-5
- 4-Hydroxy-3-(3-methyl-2-butenoyl)-5-(3-methyl-2-butenyl)benzoic acid
Catalog No.:BCN1554
CAS No.:155051-85-7
- Usaramine
Catalog No.:BCN2121
CAS No.:15503-87-4
- Isatidine
Catalog No.:BCN2120
CAS No.:15503-86-3
- Pancuronium dibromide
Catalog No.:BCC4578
CAS No.:15500-66-0
- Ac-Arg-OH.2H2O
Catalog No.:BCC2855
CAS No.:155-84-0
- Rhaponiticin
Catalog No.:BCN5392
CAS No.:155-58-8
- Methscopolamine
Catalog No.:BCC4577
CAS No.:155-41-9
- RU 58841
Catalog No.:BCC1911
CAS No.:154992-24-2
- Erysenegalensein E
Catalog No.:BCN3979
CAS No.:154992-17-3
- 6-ethyl-3-methyl-4-oxo-4H-pyran-2-carboxylic acid
Catalog No.:BCC8270
CAS No.:1551-49-1
- 2-[1-(4-Piperonyl)piperazinyl]benzothiazole
Catalog No.:BCC6771
CAS No.:155106-73-3
- Rosiglitazone maleate
Catalog No.:BCC2262
CAS No.:155141-29-0
- Plerixafor 8HCl (AMD3100 8HCl)
Catalog No.:BCC4447
CAS No.:155148-31-5
- Plerixafor octahydrobromide
Catalog No.:BCC9123
CAS No.:155148-32-6
- Physapruin A
Catalog No.:BCN7576
CAS No.:155178-03-3
- Cordifolioside A
Catalog No.:BCN8224
CAS No.:155179-20-7
- Cinnamylideneacetic acid
Catalog No.:BCN7777
CAS No.:1552-94-9
- 4-(Dimethylamino)cinnamic acid
Catalog No.:BCN5031
CAS No.:1552-96-1
- 7alpha,15-Dihydroxydehydroabietic acid
Catalog No.:BCN7672
CAS No.:155205-64-4
- Methyl 7,15-dihydroxydehydroabietate
Catalog No.:BCN1693
CAS No.:155205-65-5
- Bimatoprost
Catalog No.:BCC4948
CAS No.:155206-00-1
[Studies on triterpenes chemical constituents in rhizome of Alisma gramineum].[Pubmed:16245905]
Zhongguo Zhong Yao Za Zhi. 2005 Aug;30(16):1263-5.
OBJECTIVE: To investigate the triterpenoids chemical constituents in rhizome of Alisma gramineum. METHOD: Silicon gel chromatography and HPLC techniques were employed for isolation and purification of the constituents, and the structures were elucidated by various spectral analyses such as NMR, MS and IR. RESULT: 5 protostane-type tetracyclic triterpenoids were isolated and identified as 16,23-oxidoalisol B(1), 11-deoxyalisol C(2), alisol F(3), 16beta-methoxyalisol B acetate (4), 16beta-hydroxyalisol B acetate (5). CONCLUSION: All of these compounds were found from A. gramineum for the first time.
Structures and biological activities of the triterpenoids and sesquiterpenoids from Alisma orientale.[Pubmed:27615692]
Phytochemistry. 2016 Nov;131:150-157.
Sixteen triterpenoids and nine sesquiterpenoids were isolated from the rhizome of Alisma orientale. Structures of 16-oxo-11-anhydroalisol A 24-acetate, 13beta,17beta-epoxy-24,25,26,27-tetranor-alisol A 23-oic acid, 1alphaH,5alphaH-guaia-6-ene-4beta,10beta-diol, and alisguaiaone were elucidated by comprehensive spectroscopic data analysis. The cytotoxic, antibacterial, antifungal, anti-inflammatory, and alpha-glucosidase inhibitory activities of isolated terpenoids were evaluated. Triterpenoids alisol A, alisol A 24-acetate, 25-O-ethylalisol A, 11-deoxyalisol A, alisol E 24-acetate, alisol G, alisol B 23-acetate and sesquiterpenoids 1alphaH,5alphaH-guaia-6-ene-4beta,10beta-diol, 10-hydroxy-7,10-epoxysalvialane exhibited cytotoxicities against the three tested human cancer cell lines with IC50 values ranging from 11.5 +/- 1.7 muM to 76.7 +/- 1.4 muM. Triterpenoids alisol A, 25-O-ethylalisol A, 11-deoxyalisol A, alisol E 24-acetate, alisol G, and 25-anhydroalisol F showed antibacterial activities against the Gram-positive strains Bacillus subtilis and Staphylococcus aureus with MIC values of 12.5-100 mug/mL. Sesquiterpenoid 4beta,10beta-dihydroxy-1alphaH,5betaH-guaia-6-ene exhibited antibacterial activity against B. subtilis with an MIC value of 50 mug/mL, and 10-hydroxy-7,10-epoxysalvialane exhibited activity against S. aureus with an MIC value of 100 mug/mL. Compounds 16-oxo-11-anhydroalisol A 24-acetate, alisol F, 25-anhydroalisol F, and alisguaiaone exhibited inhibitory effects on lipopolysaccharide-induced NO production in RAW 264.7 macrophage cells. None of the compounds showed obvious inhibitory activity against alpha-glucosidase.
A new triterpenoid from Alisma orientale and their antibacterial effect.[Pubmed:23212633]
Arch Pharm Res. 2012 Nov;35(11):1919-26.
A new triterpenoid, named alisol Q 23-acetate, as well as fourteen known terpenes, alisol B 23-acetate (2), alisol B (3), alismol (4), 10-O-methyl-alismoxide (5), alismoxide (6), 11-deoxyalisol C (7), 13beta,17beta-epoxyalisol B 23-acetate (8), 4beta,12-dihydroxyguaian-6,10-diene (9), alisol C 23-acetate (10), alisolide (11), 16beta-methoxyalisol B monoacetate (12), alisol A (13), 16beta-hydroxyalisol B 23-acetate (14), alisol A 24-acetate (15) were isolated from the rhizomes of Alisma orientale. The structures of compounds (1-15) were identified based on 1D and 2D NMR, including (1)H-(1)H COSY, HSQC, HMBC and NOESY spectroscopic analyses. Among these isolates, antibacterial effect of compounds 2, 3, 10, and 15, major constituents of A. orientale was examined. The MIC values of compounds 2, 10, and 15 were 5-10 betag/mL against eight antibiotic resistant strains, which were lower than those from the positive controls (MICs of chloramphenicol and ampicillin were 5-80 mug/mL). Therefore, compounds 2, 10 and 15 exhibited the potent antibacterial activity.
Effects of sesquiterpenes and triterpenes from the rhizome of Alisma orientale on nitric oxide production in lipopolysaccharide-activated macrophages: absolute stereostructures of alismaketones-B 23-acetate and -C 23-acetate.[Pubmed:10560729]
Bioorg Med Chem Lett. 1999 Nov 1;9(21):3081-6.
The methanolic extract from a Chinese herbal medicine, the rhizome of Alisma orientale, was found to exhibit inhibitory activity of nitric oxide (NO) production in lipopolysaccharide (LPS)activated macrophages. Novel triterpenes, alismaketones-B 23-acetate and -C 23-acetate, were isolated from the active extract together with eight sesquiterpenes and eighteen protostane-type triterpenes. The absolute stereostructures of new triterpenes were characterized on the basis of chemical and physicochemical evidence, which included the chemical correlations with known triterpenes. The guaiane-type sesquiterpenes (alismol, orientalols A and C) and protostane- and seco-protostane-types triterpenes (alisols C monoacetate, E-23-acetate, F, H, I, L-23-acetate, and M-23-acetate, alismaketones-B 23-acetate and -C 23-acetate, alismalactone 23-acetate, and 3-methylalismalactone 23-acetate) inhibited LPS-induced NO production (IC50 = 8.4-68 microM). Other triterpenes (alisols A, A monoacetate, B, B monoacetate, E, G, K-23-acetate, and N-23-acetate and 11-Deoxyalisol B) also showed the potent inhibitory activity, but they showed cytotoxic effects more than 30 microM (MTT assay). In addition, alismol and alisol F were found to suppress iNOS induction.


