10-HydroxycamptothecinCAS# 64439-81-2 |
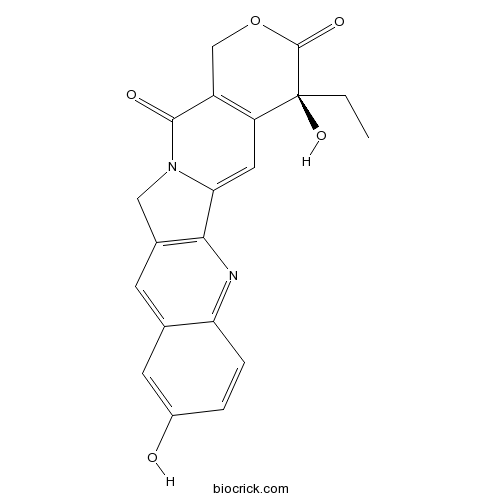
- (S)-10-Hydroxycamptothecin
Catalog No.:BCN1225
CAS No.:19685-09-7
Quality Control & MSDS
Number of papers citing our products

Chemical structure

3D structure
| Cas No. | 64439-81-2 | SDF | Download SDF |
| PubChem ID | 4330531 | Appearance | Yellow powder |
| Formula | C20H16N2O5 | M.Wt | 364.35 |
| Type of Compound | Alkaloids | Storage | Desiccate at -20°C |
| Solubility | DMSO : ≥ 37.5 mg/mL (102.92 mM) *"≥" means soluble, but saturation unknown. | ||
| Chemical Name | 19-ethyl-7,19-dihydroxy-17-oxa-3,13-diazapentacyclo[11.8.0.02,11.04,9.015,20]henicosa-1(21),2(11),3,5,7,9,15(20)-heptaene-14,18-dione | ||
| SMILES | CCC1(C2=C(COC1=O)C(=O)N3CC4=C(C3=C2)N=C5C=CC(=CC5=C4)O)O | ||
| Standard InChIKey | HAWSQZCWOQZXHI-UHFFFAOYSA-N | ||
| Standard InChI | InChI=1S/C20H16N2O5/c1-2-20(26)14-7-16-17-11(5-10-6-12(23)3-4-15(10)21-17)8-22(16)18(24)13(14)9-27-19(20)25/h3-7,23,26H,2,8-9H2,1H3 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | 10-Hydroxycamptothecin is a cell-permeable powerful DNA topoisomerase I inhibitor. It has selective inhibitory effect on the phosphorylation of histone H1 and H3, but less effect on other histones.10-Hydroxycamptothecin anticancer, and antiangiogenic activities, it can prevent fibroblast proliferation and epidural scar adhesion after laminectomy in rats. |
| Targets | DNA topoisomerase I |
| In vitro | Intracellular target delivery of 10-hydroxycamptothecin with solid lipid nanoparticles against multidrug resistance.[Pubmed: 25766079]J Drug Target. 2015 Mar 13:1-6.The main objective of this study was to design a suitable drug delivery system for 10-Hydroxycamptothecin (HCPT). Antiangiogenic potential of 10-hydroxycamptothecin.[Pubmed: 11589502]Life Sci. 2001 Aug 24;69(14):1619-28.
|
| In vivo | Preclinical pharmacology of the natural product anticancer agent 10-hydroxycamptothecin, an inhibitor of topoisomerase I.[Pubmed: 9488594 ]Cancer Chemother Pharmacol. 1998;41(4):257-67.10-Hydroxycamptothecin (HCPT) is an indole alkaloid isolated from a Chinese tree, Camptotheca acuminata, and has a wide spectrum of anticancer activity in vitro and in vivo mainly through inhibitory effects on topoisomerase I. HCPT has been shown to be more potent and less toxic than camptothecin and has recently undergone clinical trials. To determine how HCPT might be best used as an anticancer agent, preclinical studies of the pharmacokinetics, tissue distribution, metabolism and elimination of HCPT in rats were undertaken.
|
| Animal Research | The effect of 10-hydroxycamptothecine in preventing fibroblast proliferation and epidural scar adhesion after laminectomy in rats.[Pubmed: 18680742]Eur J Pharmacol. 2008 Sep 28;593(1-3):44-8.Little is known about the influence of 10-Hydroxycamptothecin (HCPT) on fibroblast proliferation and pathological changes in epidural scar tissue after laminectomy. Here we illustrated the effect of HCPT on fibroblast proliferation and epidural scar adhesion after laminectomy in rats. |

10-Hydroxycamptothecin Dilution Calculator

10-Hydroxycamptothecin Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 2.7446 mL | 13.7231 mL | 27.4461 mL | 54.8923 mL | 68.6153 mL |
| 5 mM | 0.5489 mL | 2.7446 mL | 5.4892 mL | 10.9785 mL | 13.7231 mL |
| 10 mM | 0.2745 mL | 1.3723 mL | 2.7446 mL | 5.4892 mL | 6.8615 mL |
| 50 mM | 0.0549 mL | 0.2745 mL | 0.5489 mL | 1.0978 mL | 1.3723 mL |
| 100 mM | 0.0274 mL | 0.1372 mL | 0.2745 mL | 0.5489 mL | 0.6862 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Dulcoside A
Catalog No.:BCN3237
CAS No.:64432-06-0
- Grifolin monomethyl ether
Catalog No.:BCN7568
CAS No.:64432-04-8
- Shanzhiside methylester
Catalog No.:BCN4187
CAS No.:64421-28-9
- Mussaenoside
Catalog No.:BCN4186
CAS No.:64421-27-8
- DL-2-Amino-n-octanoic acid
Catalog No.:BCC8949
CAS No.:644-90-6
- Precocene II
Catalog No.:BCN4605
CAS No.:644-06-4
- Alloalantolactone
Catalog No.:BCN8091
CAS No.:64340-41-6
- N6-Benzoyl-5'-O-(4,4'-dimethoxytrityl)-2'-deoxyadenosine
Catalog No.:BCC9074
CAS No.:64325-78-6
- Malvidin chloride
Catalog No.:BCN3017
CAS No.:643-84-5
- Dracorhodin
Catalog No.:BCC9226
CAS No.:643-56-1
- Galactopinitol A
Catalog No.:BCC8926
CAS No.:64290-91-1
- Acetagastrodin
Catalog No.:BCN8155
CAS No.:64291-41-4
- Tizanidine HCl
Catalog No.:BCC4357
CAS No.:64461-82-1
- Picroside III
Catalog No.:BCN6324
CAS No.:64461-95-6
- Glycoursodeoxycholic acid
Catalog No.:BCN7369
CAS No.:64480-66-6
- Cefotaxime sodium
Catalog No.:BCC8908
CAS No.:64485-93-4
- Rubianthraquinone
Catalog No.:BCN6880
CAS No.:644967-44-2
- Altretamine
Catalog No.:BCC1216
CAS No.:645-05-6
- Isovanillic acid
Catalog No.:BCN3376
CAS No.:645-08-9
- Scoulerine
Catalog No.:BCN6623
CAS No.:6451-73-6
- Oxprenolol hydrochloride
Catalog No.:BCC7630
CAS No.:6452-73-9
- Acantrifoside E
Catalog No.:BCN6646
CAS No.:645414-25-1
- Sinigrin monohydrate
Catalog No.:BCN2595
CAS No.:64550-88-5
- H-D-Glu(OMe)-OH
Catalog No.:BCC2940
CAS No.:6461-04-7
The effect of 10-hydroxycamptothecine in preventing fibroblast proliferation and epidural scar adhesion after laminectomy in rats.[Pubmed:18680742]
Eur J Pharmacol. 2008 Sep 28;593(1-3):44-8.
Little is known about the influence of 10-Hydroxycamptothecin (HCPT) on fibroblast proliferation and pathological changes in epidural scar tissue after laminectomy. Here we illustrated the effect of HCPT on fibroblast proliferation and epidural scar adhesion after laminectomy in rats. In the present study, seventy-two rats underwent laminectomies at Lumbar-1 level, then HCPT in various concentrations (0.1, 0.05, and 0.01 mg/ml) or saline (9 mg/ml) were applied to the laminectomy sites. Four weeks later the rats were killed and the epidural adhesion was evaluated. The area of epidural scar tissue and the number of fibroblasts were also determined. The degree of epidural adhesion was classified according to Rydell standard. The results showed that no or little epidural adhesions were seen in the laminectomy sites treated with 0.1 mg/ml HCPT. The Rydell classification, the area of epidural scar tissue and the number of fibroblasts in 0.1 mg/ml HCPT group were significantly less than those of 0.05 mg/ml HCPT group, 0.01 mg/ml HCPT group and saline group. Moderate epidural adhesions were noted in the laminectomy sites of 0.05 mg/ml HCPT group. The Rydell classification, the area of scar tissue and the number of fibroblasts were less than those of 0.01 mg/ml HCPT group and saline group. However, dense epidural adhesions were found in 0.01 mg/ml HCPT group and saline group. The Rydell classification, the area of scar tissue and the number of fibroblasts showed no significant difference compared with those of saline group. In conclusion, topical application of 0.1 mg/ml HCPT could effectively prevent fibroblast proliferation and reduce epidural adhesion after laminectomy in rats.
Preclinical pharmacology of the natural product anticancer agent 10-hydroxycamptothecin, an inhibitor of topoisomerase I.[Pubmed:9488594]
Cancer Chemother Pharmacol. 1998;41(4):257-67.
PURPOSE: 10-Hydroxycamptothecin (HCPT) is an indole alkaloid isolated from a Chinese tree, Camptotheca acuminata, and has a wide spectrum of anticancer activity in vitro and in vivo mainly through inhibitory effects on topoisomerase I. HCPT has been shown to be more potent and less toxic than camptothecin and has recently undergone clinical trials. To determine how HCPT might be best used as an anticancer agent, preclinical studies of the pharmacokinetics, tissue distribution, metabolism and elimination of HCPT in rats were undertaken. METHODS: HCPT was administered to rats by i.v. bolus injection at doses of 1, 3, and 10 mg/kg body weight. HCPT (lactone and carboxylate) and its metabolites in plasma, urine, feces, and various tissues were quantitated by reversed-phase HPLC. Pharmacokinetic parameters were then estimated. RESULTS: Following i.v. administration at doses of 3 or 10 mg/kg, the plasma concentration-time profile for lactone HCPT could be best described by a three-compartment model, with terminal elimination half-lives of 140.4 and 428.6 min, respectively. A two-compartment model was used to fit the plasma concentration-time curve at 1 mg/kg, with a terminal elimination half-life of 30.5 min. Carboxylate HCPT had a longer half-life than the lactone form of HCPT. During the initial 6 h after dosing, urinary excretion was the major route of elimination, and fecal excretion became the major route of elimination thereafter. HCPT was widely distributed to various tissues including the enterohepatic system, kidney, and bone marrow. The lactone form of HCPT was detectable in various tissues examined up to 72 h after dosing at all the three test doses. HCPT glucuronides were present in plasma, urine, feces and various tissues. No significant toxicity was observed at doses of 1 or 3 mg/kg. Polyuria and hematuria were observed only during the initial 3 h after dosing at 10 mg/kg. CONCLUSIONS: Prolonged elimination of HCPT in vivo may have a significant impact on its therapeutic effects. HCPT is metabolized to its carboxylate form and glucuronides. Dose-dependent toxicity was observed with i.v. administration of HCPT. The results of this study should be useful in the design of future human trials with this anticancer drug.
Intracellular target delivery of 10-hydroxycamptothecin with solid lipid nanoparticles against multidrug resistance.[Pubmed:25766079]
J Drug Target. 2015;23(9):800-5.
The main objective of this study was to design a suitable drug delivery system for 10-Hydroxycamptothecin (HCPT). In this study, HCPT-loaded solid lipid nanoparticle (HCPT-loaded SLN) was successfully prepared. The HCPT-loaded SLN was characterized by size, entrapment efficiency and drug release manner. The cytotoxicity of HCPT-loaded SLN was assessed in vitro using HepG2/HCPT cells and in vivo utilizing human tumor xenograft nude mouse model. HCPT-loaded SLN indicated the ability to target HepG2/HCPT cells and accumulated higher drug content in HepG2/HCPT cells. HCPT-loaded SLN enhanced the cytotoxicity of HCPT in a concentration-dependent manner. Based on these results, HCPT-loaded SLN suggested being a promising vehicle for liver-targeted drug delivery. Moreover, it can be of clinical interest to overcome multidrug resistance (MDR) effectively.
Antiangiogenic potential of 10-hydroxycamptothecin.[Pubmed:11589502]
Life Sci. 2001 Aug 24;69(14):1619-28.
To investigate the antiangiogenic potential of 10-Hydroxycamptothecin (HCPT), the proliferation of human microvascular endothelial cells (HMEC) and seven human tumor cell lines were detected by SRB assay, and the endothelial cell migration and tube formation were assessed using two in vitro model systems. Also, inhibition of angiogenesis was determined with a modification of the chick embryo chorioallantoic membrane (CAM) assay in vivo. Morphological assessment of apoptosis was performed by fluorescence microscope. HCPT 0.313-5 micromol x L(-1) treatment resulted in a dose-dependent inhibition of proliferation, migration and tube formation in HMEC cells, and HCPT 6.25-25 nmol x egg(-1) inhibited angiogenesis in CAM assay. HCPT 1.25-5 micromol x L(-1) elicited typical morphological changes of apoptosis including condensed chromatin, nuclear fragmentation, and reduction in volume in HMEC cells. HCPT significantly inhibited angiogenesis both in vitro and in vivo at relatively low concentrations, and this effect was related with induction of apoptosis in HMEC cells. These results taken collectively suggest that HCPT may be a potent antiangiogenetic and cytotoxic drug and further investigation is warranted.


