1-EBIOKCa activator; promotes ESC differentiation into cardiomyocytes CAS# 10045-45-1 |
- CYT387 sulfate salt
Catalog No.:BCC1506
CAS No.:1056636-06-6
- Baricitinib phosphate
Catalog No.:BCC1401
CAS No.:1187595-84-1
- JAK2 Inhibitor V, Z3
Catalog No.:BCC1667
CAS No.:195371-52-9
- Bardoxolone methyl
Catalog No.:BCC1400
CAS No.:218600-53-4
- Ruxolitinib (INCB018424)
Catalog No.:BCC1276
CAS No.:941678-49-5
Quality Control & MSDS
Number of papers citing our products

Chemical structure

3D structure
| Cas No. | 10045-45-1 | SDF | Download SDF |
| PubChem ID | 82320 | Appearance | Powder |
| Formula | C9H10N2O | M.Wt | 162.19 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Solubility | Soluble to 100 mM in ethanol and to 100 mM in DMSO | ||
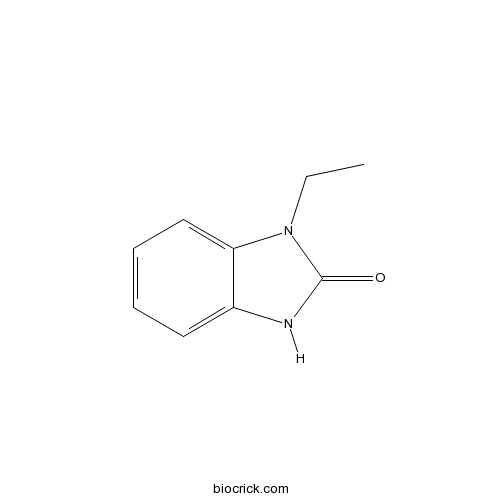
| Chemical Name | 3-ethyl-1H-benzimidazol-2-one | ||
| SMILES | CCN1C2=CC=CC=C2NC1=O | ||
| Standard InChIKey | CXUCKELNYMZTRT-UHFFFAOYSA-N | ||
| Standard InChI | InChI=1S/C9H10N2O/c1-2-11-8-6-4-3-5-7(8)10-9(11)12/h3-6H,2H2,1H3,(H,10,12) | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Activator of epithelial KCa channels, stimulates a large and sustained trans-epithelial Cl- secretory response across T84 monolayers. Induces hyperpolarization to the same magnitude as ACh in aortic value endothelial cells. Promotes embryonic stem cell (ESC) differentiation into cardiomyocytes. |

1-EBIO Dilution Calculator

1-EBIO Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 6.1656 mL | 30.828 mL | 61.6561 mL | 123.3122 mL | 154.1402 mL |
| 5 mM | 1.2331 mL | 6.1656 mL | 12.3312 mL | 24.6624 mL | 30.828 mL |
| 10 mM | 0.6166 mL | 3.0828 mL | 6.1656 mL | 12.3312 mL | 15.414 mL |
| 50 mM | 0.1233 mL | 0.6166 mL | 1.2331 mL | 2.4662 mL | 3.0828 mL |
| 100 mM | 0.0617 mL | 0.3083 mL | 0.6166 mL | 1.2331 mL | 1.5414 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Dihydroresveratrol 3-O-glucoside
Catalog No.:BCN5821
CAS No.:100432-87-9
- Cobicistat (GS-9350)
Catalog No.:BCC2271
CAS No.:1004316-88-4
- Boric acid
Catalog No.:BCC7592
CAS No.:10043-35-3
- Lercanidipine
Catalog No.:BCC5239
CAS No.:100427-26-7
- Danshinspiroketallactone
Catalog No.:BCN3754
CAS No.:100414-80-0
- Sodium Picosulfate
Catalog No.:BCC4845
CAS No.:10040-45-6
- GSK 1562590 hydrochloride
Catalog No.:BCC8010
CAS No.:1003878-07-6
- Calcium chloride dihydrate
Catalog No.:BCC7582
CAS No.:10035-04-8
- Curcumenone
Catalog No.:BCN3008
CAS No.:100347-96-4
- L(+)-Rhamnose monohydrate
Catalog No.:BCN8368
CAS No.:10030-85-0
- Pemirolast potassium
Catalog No.:BCC4532
CAS No.:100299-08-9
- Apiopaeonoside
Catalog No.:BCN2801
CAS No.:100291-86-9
- FFN 511
Catalog No.:BCC7799
CAS No.:1004548-96-2
- Rosiridin
Catalog No.:BCN5970
CAS No.:100462-37-1
- Gastrin I (human)
Catalog No.:BCC5958
CAS No.:10047-33-3
- Sterigmatocystin
Catalog No.:BCN6885
CAS No.:10048-13-2
- Blasticidin A
Catalog No.:BCN1830
CAS No.:100513-53-9
- TCS 2002
Catalog No.:BCC6074
CAS No.:1005201-24-0
- Aeruginolactone
Catalog No.:BCN3695
CAS No.:1005208-88-7
- Gelomulide N
Catalog No.:BCN6641
CAS No.:1005212-02-1
- CVT 10216
Catalog No.:BCC5606
CAS No.:1005334-57-5
- LCL161
Catalog No.:BCC1691
CAS No.:1005342-46-0
- Tirasemtiv
Catalog No.:BCC5183
CAS No.:1005491-05-3
- Monomethylsulochrin
Catalog No.:BCN7255
CAS No.:10056-14-1
In vivo characterisation of the small-conductance KCa (SK) channel activator 1-ethyl-2-benzimidazolinone (1-EBIO) as a potential anticonvulsant.[Pubmed:16925994]
Eur J Pharmacol. 2006 Sep 28;546(1-3):48-53.
Owing to their activation by increased intracellular Ca(2+) levels following burst firing, and the resultant hyperpolarisation and dampening of neuronal excitability, the small-conductance Ca(2+)-activated K(+) (SK(Ca)) channels have been proposed as a potential target for novel antiepileptic drugs. Indeed, the channel activator 1-ethyl-2-benzimidazolinone (1-EBIO) has been shown to reduce epileptiform activity in vitro. Accordingly, this study has investigated the therapeutic potential of 1-EBIO using a range of in vivo seizure models, and assessed the adverse effect liability with the rotarod and locomotor activity paradigms. To aid benchmarking of 1-EBIO's therapeutic and adverse effect potential, it was tested alongside two currently marketed antiepileptic drugs, phenytoin and levetiracetam. 1-EBIO was found to be effective at reducing seizure incidence in mice following maximal electroshock (ED(50) 36.0 mg/kg) as well as increasing the threshold to electrically- and pentylenetetrazole-induced seizures (TID(10)s 7.3 and 21.5 mg/kg, respectively). However, results from the mouse rotarod test revealed a strong adverse effect potential within the therapeutic dose range (ID(50) 35.6 mg/kg), implying a significantly inferior therapeutic index with respect to the comparator compounds. These results, therefore, support the in vitro data detailing 1-EBIO's reduction of epileptiform activity. However, the use of in vivo models has revealed a significant adverse effect potential within the therapeutic dose range. Nevertheless, given the multiplicity of SK(Ca) channel subunits and that 1-EBIO has been shown to enhance additional, non-SK(Ca) carried currents, these findings do not preclude the possibility that more selective enhancers of SK(Ca) function could prove to be effective as antiepileptic medications.
Activation of K(+) channel by 1-EBIO rescues the head and neck squamous cell carcinoma cells from Ca(2+) ionophore-induced cell death.[Pubmed:26807020]
Korean J Physiol Pharmacol. 2016 Jan;20(1):25-33.
Ion channels in carcinoma and their roles in cell proliferation are drawing attention. Intracellular Ca(2+) ([Ca(2+)]i)-dependent signaling affects the fate of cancer cells. Here we investigate the role of Ca(2+)-activated K(+) channel (SK4) in head and neck squamous cell carcinoma cells (HNSCCs) of different cell lines; SNU-1076, OSC-19 and HN5. Treatment with 1 microM ionomycin induced cell death in all the three cell lines. Whole-cell patch clamp study suggested common expressions of Ca(2+)-activated Cl(-) channels (Ano-1) and Ca(2+)-activated nonselective cation channels (CAN). 1-EBIO, an activator of SK4, induced outward K(+) current (ISK4) in SNU-1076 and OSC-19. In HN5, ISK4 was not observed or negligible. The 1-EBIO-induced current was abolished by TRAM-34, a selective SK4 blocker. Interestingly, the ionomycin-induced cell death was effectively prevented by 1-EBIO in SNU-1076 and OSC-19, and the rescue effect was annihilated by combined TRAM-34. Consistent with the lower level of ISK4, the rescue by 1-EBIO was least effective in HN5. The results newly demonstrate the role of SK4 in the fate of HNSCCs under the Ca(2+) overloaded condition. Pharmacological modulation of SK4 might provide an intriguing novel tool for the anti-cancer strategy in HNSCC.
The K+ channel opener 1-EBIO potentiates residual function of mutant CFTR in rectal biopsies from cystic fibrosis patients.[Pubmed:21909392]
PLoS One. 2011;6(8):e24445.
BACKGROUND: The identification of strategies to improve mutant CFTR function remains a key priority in the development of new treatments for cystic fibrosis (CF). Previous studies demonstrated that the K(+) channel opener 1-ethyl-2-benzimidazolone (1-EBIO) potentiates CFTR-mediated Cl(-) secretion in cultured cells and mouse colon. However, the effects of 1-EBIO on wild-type and mutant CFTR function in native human colonic tissues remain unknown. METHODS: We studied the effects of 1-EBIO on CFTR-mediated Cl(-) secretion in rectal biopsies from 47 CF patients carrying a wide spectrum of CFTR mutations and 57 age-matched controls. Rectal tissues were mounted in perfused micro-Ussing chambers and the effects of 1-EBIO were compared in control tissues, CF tissues expressing residual CFTR function and CF tissues with no detectable Cl(-) secretion. RESULTS: Studies in control tissues demonstrate that 1-EBIO activated CFTR-mediated Cl(-) secretion in the absence of cAMP-mediated stimulation and potentiated cAMP-induced Cl(-) secretion by 39.2+/-6.7% (P<0.001) via activation of basolateral Ca(2)(+)-activated and clotrimazole-sensitive KCNN4 K(+) channels. In CF specimens, 1-EBIO potentiated cAMP-induced Cl(-) secretion in tissues with residual CFTR function by 44.4+/-11.5% (P<0.001), but had no effect on tissues lacking CFTR-mediated Cl(-) conductance. CONCLUSIONS: We conclude that 1-EBIO potentiates Cl(-)secretion in native CF tissues expressing CFTR mutants with residual Cl(-) channel function by activation of basolateral KCNN4 K(+) channels that increase the driving force for luminal Cl(-) exit. This mechanism may augment effects of CFTR correctors and potentiators that increase the number and/or activity of mutant CFTR channels at the cell surface and suggests KCNN4 as a therapeutic target for CF.
Chlorzoxazone or 1-EBIO increases Na(+) absorption across cystic fibrosis airway epithelial cells.[Pubmed:11597903]
Am J Physiol Lung Cell Mol Physiol. 2001 Nov;281(5):L1123-9.
Previous studies demonstrated that chlorzoxazone or 1-ethyl-2-benzimidazolinone (1-EBIO) enhances transepithelial Cl(-) secretion by increasing basolateral K(+) conductance (G(K)) (Singh AK, Devor DC, Gerlach AC, Gondor M, Pilewski JM, and Bridges RJ. J Pharmacol Exp Ther 292: 778-787, 2000). Hence these compounds may be useful to treat cystic fibrosis (CF) airway disease. The goal of the present study was to determine whether chlorzoxazone or 1-EBIO altered ion transport across Delta F508-CF transmembrane conductance regulator homozygous CFT1 airway cells. CFT1 monolayers exhibited a basal short-circuit current that was abolished by apical amiloride (inhibition constant 320 nM) as expected for Na(+) absorption. The addition of chlorzoxazone (400 microM) or 1-EBIO (2 mM) increased the amiloride-sensitive I(sc) approximately 2.5-fold. This overlapping specificity may preclude use of these compounds as CF therapeutics. Assaying for changes in the basolateral G(K) with a K(+) gradient plus the pore-forming antibiotic amphotericin B revealed that chlorzoxazone or 1-EBIO evoked an approximately 10-fold increase in clotrimazole-sensitive G(K). In contrast, chlorzoxazone did not alter epithelial Na(+) channel-mediated currents across basolateral-permeabilized monolayers or in Xenopus oocytes. These data further suggest that alterations in basolateral G(K) alone can modulate epithelial Na(+) transport.
Modulation of calcium-activated potassium channels induces cardiogenesis of pluripotent stem cells and enrichment of pacemaker-like cells.[Pubmed:20956206]
Circulation. 2010 Nov 2;122(18):1823-36.
BACKGROUND: Ion channels are key determinants for the function of excitable cells, but little is known about their role and involvement during cardiac development. Earlier work identified Ca(2+)-activated potassium channels of small and intermediate conductance (SKCas) as important regulators of neural stem cell fate. Here we have investigated their impact on the differentiation of pluripotent cells toward the cardiac lineage. METHODS AND RESULTS: We have applied the SKCa activator 1-ethyl-2-benzimidazolinone on embryonic stem cells and identified this particular ion channel family as a new critical target involved in the generation of cardiac pacemaker-like cells: SKCa activation led to rapid remodeling of the actin cytoskeleton, inhibition of proliferation, induction of differentiation, and diminished teratoma formation. Time-restricted SKCa activation induced cardiac mesoderm and commitment to the cardiac lineage as shown by gene regulation, protein, and functional electrophysiological studies. In addition, the differentiation into cardiomyocytes was modulated in a qualitative fashion, resulting in a strong enrichment of pacemaker-like cells. This was accompanied by induction of the sino-atrial gene program and in parallel by a loss of the chamber-specific myocardium. In addition, SKCa activity induced activation of the Ras-Mek-Erk signaling cascade, a signaling pathway involved in the 1-ethyl-2-benzimidazolinone-induced effects. CONCLUSIONS: SKCa activation drives the fate of pluripotent cells toward mesoderm commitment and cardiomyocyte specification, preferentially into nodal-like cardiomyocytes. This provides a novel strategy for the enrichment of cardiomyocytes and in particular, the generation of a specific subtype of cardiomyocytes, pacemaker-like cells, without genetic modification.
Reduced hyperpolarization in endothelial cells of rabbit aortic valve following chronic nitroglycerine administration.[Pubmed:16056232]
Br J Pharmacol. 2005 Oct;146(4):487-97.
This study was undertaken to determine whether long-term in vivo administration of nitroglycerine (NTG) downregulates the hyperpolarization induced by acetylcholine (ACh) in aortic valve endothelial cells (AVECs) of the rabbit and, if so, whether antioxidant agents can normalize this downregulated hyperpolarization. ACh (0.03-3 microM) induced a hyperpolarization through activations of both apamin- and charybdotoxin-sensitive Ca2+-activated K+ channels (K(Ca)) in rabbit AVECs. The intermediate-conductance K(Ca) channel (IK(Ca)) activator 1-ethyl-2-benzimidazolinone (1-EBIO, 0.3 mM) induced a hyperpolarization of the same magnitude as ACh (3 microM). The ACh-induced hyperpolarization was significantly weaker, although the ACh-induced [Ca2+]i increase was unchanged, in NTG-treated rabbits (versus NTG-untreated control rabbits). The hyperpolarization induced by 1-EBIO was also weaker in NTG-treated rabbits. The reduced ACh-induced hyperpolarization seen in NTG-treated rabbits was not modified by in vitro application of the superoxide scavengers Mn-TBAP, tiron or ascorbate, but it was normalized when ascorbate was coadministered with NTG in vivo. Superoxide production within the endothelial cell (estimated by ethidium fluorescence) was increased in NTG-treated rabbits and this increased production was normalized by in vivo coadministration of ascorbate with the NTG. It is suggested that long-term in vivo administration of NTG downregulates the ACh-induced hyperpolarization in rabbit AVECs, possibly through chronic actions mediated by superoxide.
1-Ethyl-2-benzimidazolinone stimulates endothelial K(Ca) channels and nitric oxide formation in rat mesenteric vessels.[Pubmed:10497901]
Eur J Pharmacol. 1999 Aug 27;379(2-3):151-9.
Hyperpolarization of most blood vessels occurs by the opening of K(Ca) channels. 1-Ethyl-2-benzimidazolinone (1-EBIO) is a direct activator of K(Ca) channels in epithelial cells and is potentially valuable for studying cellular hyperpolarization. This study reports the effects of 1-EBIO on isolated rat mesenteric beds perfused with normal (4.7 mM), or high (20 or 80 mM) K+ physiological salt solution (PSS) and constricted with an alpha1-adrenoceptor agonist, cirazoline (0.3-1 microM). Arterial perfusion pressures were decreased by 1-EBIO (0.1-30 nmol) in a dose- and endothelium-dependent manner. Infusion of penitrem A (100 nM), a maxi-K+ channel blocker, or apamin (0.5 microM), a small-conductance (SK(Ca)) K+ channel blocker, produced significant increases in cirazoline-mediated tone (mm Hg): 103.3 +/- 8.7 (control) vs. 156.3 +/- 14.3 (penitrem A); or 93.0 +/- 15.8 (control) vs. 114.0 +/- 15.4 (apamin). 1-EBIO relaxations were attenuated by penitrem A, while apamin, dendrotoxin (50 nM; a Kv channel antagonist), or ouabain (100 microM; a sodium pump blocker) failed to alter the responses. I-EBIO-mediated relaxations decreased significantly with increasing extracellular [K+]: relaxations to 30 nmol were 89.3% +/- 3.2% (4.7 mM K+, normal PSS) vs. 59.5% +/- 3.4% and 19.0% +/- 3.9% for 20 and 80 mM K+ PSS, respectively. Nomega-nitro-L-arginine-methyl ester (L-NAME; 100 microM), and 1H-[1,2,4]oxadiazolo[4,3-a]quinoxalin-1-one (ODQ; 10 microM), selective inhibitors of nitric oxide synthase, and nitric oxide-sensitive guanylate cyclase, respectively, abolished 1-EBIO relaxations in vessels perfused with 20 or 80 mM K+ PSS. We conclude that: (1) maxi-K+ and SK(Ca) channels are present in rat mesenteric arterial vessels and actively contribute to vascular tone, (2) vasodilator action of 1-EBIO involves the opening of endothelial maxi-K+ channels and nitric oxide synthesis.
Modulation of Cl- secretion by benzimidazolones. I. Direct activation of a Ca(2+)-dependent K+ channel.[Pubmed:8944721]
Am J Physiol. 1996 Nov;271(5 Pt 1):L775-84.
We evaluated the effects of the novel benzimidazolone, 1-ethyl-2-benzimidazolinone (1-EBIO), on Cl- secretion across T84 monolayers. 1-EBIO stimulated a sustained Cl- secretory response at a half-maximal effective concentration of 490 microM. Charybdotoxin (CTX) inhibited the 1-EBIO-induced short-circuit current (Isc) with an inhibitory constant (Ki) of 3.6 nM, whereas 293B, an inhibitor of adenosine 3',5'-cyclic monophosphate-activated K+ channels, had no effect on the current induced by 1-EBIO. In contrast, CTX failed to inhibit the 293B-sensitive forskolin-induced Isc. The above results suggested that 1-EBIO may be activating the basolateral membrane Ca(2+)-dependent K+ channel (KCa) in these cells. This was further confirmed using nystatin to permeabilize the apical membrane in the presence of a mucosa-to-serosa K+ gradient and determining the effects of 1-EBIO on the basolateral K+ current (IK). Under these conditions, 1-EBIO induced a large increase in IK that was blocked by CTX. In membrane vesicles prepared from T84 cells, 1-EBIO stimulated 86Rb+ uptake in a CTX-sensitive manner; the Ki for inhibition by CTX was 3.5 nM. Similar to our intact monolayer studies, this 86Rb+ uptake was not blocked by 293B. The effects of 1-EBIO on the KCa in T84 cells was determined in excised inside-out patches. 1-EBIO (100 microM) increased the product of the number of channels and the open channel probability from 0.09 +/- 0.03 to 1.17 +/- 0.27 (n = 8); this effect on KCa activity required a minimal level of free Ca2+. Similar to its effect on T84 cells, 1-EBIO stimulated a sustained Cl- secretory current in rat colonic epithelium, which was partially blocked by CTX. Finally, 1-EBIO stimulated a sustained Cl- secretory response in primary cultures of murine tracheal epithelium. We conclude that the benzimidazolone, 1-EBIO, stimulates Cl- secretion in secretory epithelia via the direct activation of a Kca. 1-EBIO is the first pharmacological opener of this important class of epithelial K+ channels to be identified.


