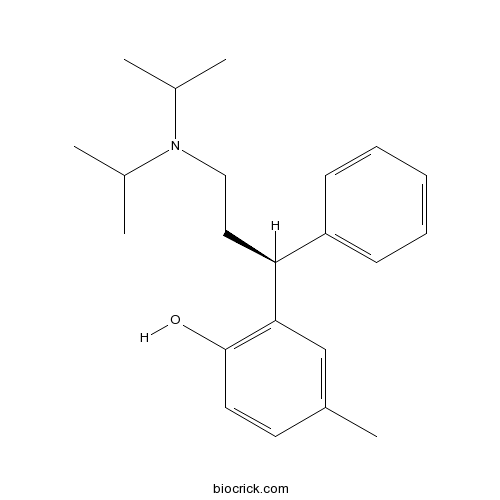
(R)-(+)-TolterodineCAS# 124937-51-5 |
- Salmefamol
Catalog No.:BCC1919
CAS No.:18910-65-1
- Guanfacine hydrochloride
Catalog No.:BCC1609
CAS No.:29110-48-3
- Doxazosin Mesylate
Catalog No.:BCC1257
CAS No.:77883-43-3
- Medetomidine
Catalog No.:BCC1736
CAS No.:86347-14-0
Quality Control & MSDS
Number of papers citing our products

Chemical structure

3D structure
| Cas No. | 124937-51-5 | SDF | Download SDF |
| PubChem ID | 60774 | Appearance | Powder |
| Formula | C22H31NO | M.Wt | 325.49 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Synonyms | (R)-(+)-Tolterodine; (+)-Tolterodine; (R)-Tolterodine; PNU-200583 | ||
| Solubility | DMSO : 250 mg/mL (768.07 mM; Need ultrasonic) | ||
| Chemical Name | 2-[(1S)-3-[di(propan-2-yl)amino]-1-phenylpropyl]-4-methylphenol | ||
| SMILES | CC1=CC(=C(C=C1)O)C(CCN(C(C)C)C(C)C)C2=CC=CC=C2 | ||
| Standard InChIKey | OOGJQPCLVADCPB-FQEVSTJZSA-N | ||
| Standard InChI | InChI=1S/C22H31NO/c1-16(2)23(17(3)4)14-13-20(19-9-7-6-8-10-19)21-15-18(5)11-12-22(21)24/h6-12,15-17,20,24H,13-14H2,1-5H3/t20-/m0/s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Tolterodine(PNU-200583) is a potent muscarinic receptor antagonists that show selectivity for the urinary bladder over salivary glands in vivo.
IC50 Value:
Target: mAChR
in vitro: Carbachol-induced contractions of isolated guinea pig bladder were effectively inhibited by tolterodine (IC50 14 nM) and 5-HM (IC50 5.7 nM). The IC50 values were in the microM range and the antimuscarinic potency of tolterodine was 27, 200 and 370-485 times higher, respectively, than its potency in blocking histamine receptors, alpha-adrenoceptors and calcium channels. The active metabolite, 5-HM, was >900 times less potent at these sites than at bladder muscarinic receptors [1].
in vivo: Tolterodine was extensively metabolized in vivo [2]. In the passive-avoidance test, tolterodine at 1 or 3 mg/kg had no effect on memory; the latency to cross and percentage of animals crossing were comparable to controls. In contrast, scopolamine induced a memory deficit; the latency to cross was decreased, and the number of animals crossing was increased [3]. References: | |||||

(R)-(+)-Tolterodine Dilution Calculator

(R)-(+)-Tolterodine Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 3.0723 mL | 15.3615 mL | 30.7229 mL | 61.4458 mL | 76.8073 mL |
| 5 mM | 0.6145 mL | 3.0723 mL | 6.1446 mL | 12.2892 mL | 15.3615 mL |
| 10 mM | 0.3072 mL | 1.5361 mL | 3.0723 mL | 6.1446 mL | 7.6807 mL |
| 50 mM | 0.0614 mL | 0.3072 mL | 0.6145 mL | 1.2289 mL | 1.5361 mL |
| 100 mM | 0.0307 mL | 0.1536 mL | 0.3072 mL | 0.6145 mL | 0.7681 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
Tolterodine is a potent muscarinic receptor antagonists that show selectivity for the urinary bladder over salivary glands in vivo.
- Benzoyl leuco methylene blue
Catalog No.:BCC8862
CAS No.:1249-97-4
- Isoaltholactone
Catalog No.:BCN4826
CAS No.:124868-11-7
- 5-Dehydroxyparatocarpin K
Catalog No.:BCN4017
CAS No.:124858-37-3
- Nadifloxacin
Catalog No.:BCC4804
CAS No.:124858-35-1
- Valacyclovir hydrochloride
Catalog No.:BCC4051
CAS No.:124832-27-5
- Valaciclovir
Catalog No.:BCC2025
CAS No.:124832-26-4
- USP7-USP47 inhibitor
Catalog No.:BCC4113
CAS No.:1247825-37-1
- P 22077
Catalog No.:BCC3616
CAS No.:1247819-59-5
- Losartan Potassium (DuP 753)
Catalog No.:BCC1080
CAS No.:124750-99-8
- Quinovic acid 3-O-(6-deoxy-beta-D-glucopyranoside) 28-O-beta-D-glucopyranosyl ester
Catalog No.:BCN1595
CAS No.:124727-10-2
- Meprednisone
Catalog No.:BCC4893
CAS No.:1247-42-3
- Methyl-Dodovisate A
Catalog No.:BCN4719
CAS No.:1246937-33-6
- Tolterodine tartrate
Catalog No.:BCC4586
CAS No.:124937-52-6
- Tectoroside
Catalog No.:BCN6672
CAS No.:124960-89-0
- Raddeanoside R8
Catalog No.:BCN6555
CAS No.:124961-61-1
- Ajugacumbin A
Catalog No.:BCN3657
CAS No.:124961-66-6
- Ajugamarin L2
Catalog No.:BCN3662
CAS No.:124961-67-7
- Prednisone 21-acetate
Catalog No.:BCC9128
CAS No.:125-10-0
- Isobornyl acetate
Catalog No.:BCN8296
CAS No.:125-12-2
- Vomicine
Catalog No.:BCN6735
CAS No.:125-15-5
- Primidone
Catalog No.:BCC4930
CAS No.:125-33-7
- Usnic acid
Catalog No.:BCC8264
CAS No.:125-46-2
- Aminoglutethimide
Catalog No.:BCC4368
CAS No.:125-84-8
- 8-Prenylluteone
Catalog No.:BCN4771
CAS No.:125002-91-7
Enantiomeric separation of R,S-tolterodine and R,S-methoxytolterodine with negatively charged cyclodextrins by capillary electrophoresis.[Pubmed:23424041]
J Sep Sci. 2013 May;36(9-10):1561-7.
The methods for separation of R,S-tolterodine and R,S-methoxytolterodine enantiomers using sulfated alpha-, beta-CD and phosphated-gamma-CD by CE in acidic BGE based on Tris/phosphate pH 2.5 buffer were developed. Sulfated alpha- and beta-CD allow anodic detection while phosphated-gamma-CD allows only cathodic detection of the separated enantiomers. The influence of chiral selector (CS)'s concentration as well as the influence of composition and concentration of BGE on resolutions were studied. Reversal migration order of tolterodine and methoxytolterodine enantiomers was observed, when sulfated-alpha- and sulfated-beta-CD were used. The developed methods with all three studied CSs, were validated and compared. All proposed methods enable determination of 0.2% of S-tolterodine as an optical impurity in pills, however the method with phosphated-gamma-CD provided lower detection limit, better repeatability of peak areas and migration times, and also lower consumption of CS. Developed method employing phosphated-gamma-CD that was applied for the determination of optical purity of R-tolterodine in commercial pills.
Enantioselective synthesis of (R)-tolterodine using lithiation/borylation-protodeboronation methodology.[Pubmed:23750041]
Can J Chem. 2012 Nov;90(11):965.
The synthesis of the pharmaceutical (R)-tolterodine is reported using lithiation/borylation-protodeboronation of a homoallyl carbamate as the key step. This step was tested with two permutations: an electron-neutral aryl Li-carbamate reacting with an electron-rich boronic ester and an electron-rich aryl Li-carbamate reacting with an electron-neutral boronic ester. It was found that the latter arrangement was considerably better than the former. Further improvements were achieved using magnesium bromide in methanol leading to a process that gave high yield and high enantioselectivity in the lithiation/borylation reaction. The key step was used in an efficient synthesis of (R)-tolterodine in a total of eight steps in a 30% overall yield and 90% ee.
Coumarins from free ortho-hydroxy cinnamates by Heck-Matsuda arylations: a scalable total synthesis of (R)-tolterodine.[Pubmed:23190249]
Org Lett. 2012 Dec 7;14(23):6036-9.
Free ortho-hydroxy cinnamate ester derivatives are evaluated in the synthesis of structurally diverse 4-aryl-coumarins via a tandem Heck-Matsuda cyclization reaction. Free phenolic groups were considered incompatible with such a reaction, which usually provide the corresponding diazo dyes. A concise and scalable route employing a ligand-free, Pd-catalyzed Heck-Matsuda arylation under aerobic conditions for the preparation of (R)-Tolterodine in high overall yield and ee is also presented.


